Associate Director, Pharmacy Services - Medication Safety and Quality
UI Health
Clinical Assistant Professor
University of Illinois Chicago Retzky College of Pharmacy
Identifying and implementing best practices is a fundamental principle of performance improvement in healthcare. These best practices evolve continuously, especially with the advent of new technologies.
When a new technology is deemed a best practice, it is typically assumed that it will be adopted and implemented as designed. However, in real-world settings, end users often develop methods of interacting with new technologies that may undermine or completely circumvent the safety benefits these systems are intended to provide. Such improvised and unsanctioned workflows are commonly referred to as “workarounds.”1 This article explores the nature of workarounds and examines specific examples related to the relatively new best-practice technologies in pharmacy known as IV workflow management systems (IVWMS).
Exploring Workarounds
The term “workaround” lacks a standardized definition; various interpretations exist, each carrying its own industry-specific connotation. For patient safety professionals, workarounds often evoke a negative response because they typically indicate a deviation from processes designed to enhance patient safety. Conversely, front-line staff may view workarounds as pragmatic solutions to cope with production pressures or optimize workflow efficiency. In this article, we adopt the perspective of patient safety while acknowledging the real-world experiences of front-line staff.
IVWMS Elements
IVWMS encompass a suite of technologies aimed at streamlining and automating pharmacy workflows that have traditionally relied on human intervention, making them susceptible to errors.2 Key components of an IVWMS may include3:
- computerized product identification (eg, barcode scanning);
- automated calculations;
- assignment of beyond-use dates;
- prospective image capture of IV components (eg, diluent and drug via lVs, syringe volumes, etc);
- gravimetric confirmation of the prepared products;
- electronic quarantine of products pending final verification;
- addressing workarounds: application to IVWMS; and
- standardization of workflow.
Standard operating procedures (SOPs) are essential for setting clear expectations, minimizing variability and facilitating the training of new staff. In the context of IVWMS, standardization should encompass both the IV preparation workflow and associated processes. For example, SOPs for new product onboarding should require that new product barcodes are programmed to align with the existing order catalog and formulary. This is particularly important in the current era of common drug shortages. Moreover, SOPs should outline specific barcode scanning protocols, highlighting pitfalls such as scanning a single vial multiple times instead of all relevant vials, or scanning vials retrospectively.
Furthermore, SOPs should outline which preparation elements require photographic documentation, ensuring front-line staff understand the need for clarity and thoroughness. Depending on the specific design of the IVWMS, process owners should determine whether/when reconstitution vials, administration sets/tubing, or final labeled preparations require a photo to be taken by the end user.
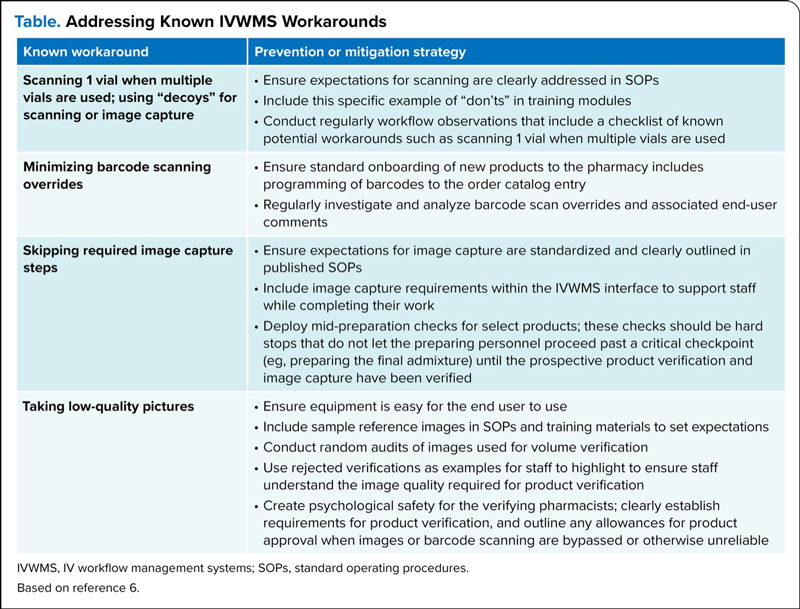
| Table. Addressing Known IVWMS Workarounds | |
| Known workaround | Prevention or mitigation strategy |
|---|---|
| Scanning 1 vial when multiple vials are used; using “decoys” for scanning or image capture |
|
| Minimizing barcode scanning overrides |
|
| Skipping required image capture steps |
|
| Taking low-quality pictures |
|
| IVWMS, IV workflow management systems; SOPs, standard operating procedures. Based on reference 6. | |
Sensitivity to Operations
Sensitivity to operations is 1 of the 5 principles characterizing highly reliable organizations.4 In healthcare, this principle underscores the importance of comprehending the complex nature of patient care workflows and how new processes or technologies may affect efficiency. Workarounds often arise when standardized processes create operational inefficiencies. In the context of IVWMS, this requires a careful examination of each workflow step to ensure it adds value. For instance, if barcode scanning verifies medication products, it may be reasonable to forgo additional image capture requirements for scanned items. Considerations also might be made to limit the number or type of products (eg, high-alert medications) that require gravimetric product confirmation. By remaining sensitive to operations, organizations can demonstrate end-user empathy and enhance the practicality of prescribed workflows without sacrificing safety, thereby reducing the likelihood of workarounds.
Anticipating and Responding to Drift
The concept of “drift” is integral to the just culture framework in patient safety. Drift refers to a gradual shift from standardized practices when deviations are neither recognized nor corrected. Although individual changes may seem insignificant, accumulated drift can lead to substantial safety risks.5 In IVWMS, drift may manifest at any process node, such as overlooking barcode overrides and relying on human verification. Drift also may arise as a failure to capture all required images during preparation (eg, only taking a picture of the drug volume in the syringe but not the diluent volume used for reconstitution). However, the drift could be more subtle, such as a pharmacist’s verification of preparations compared against images of syringes that are not of sufficient quality to confirm volumes. Organizations can counteract drift by fostering a culture of reporting near misses, regularly observing workflows, and analyzing barcode override logs and the associated end-user comments. If technically possible, blind analyses can be considered, where verifiers input observed volumes without prior knowledge of expected outcomes. Blind analysis can mitigate confirmation bias and ensure robust product verification less susceptible to drift.
Conclusion
Like all advancements in the medication-use process, IVWMS are vulnerable to workarounds. Strategies such as workflow standardization, operational sensitivity, and proactive measures against drift can effectively address and mitigate workarounds, preserving the safety benefits these systems are designed to offer.
Dr. Bursua reported no relevant financial disclosures.
References
- Boonstra A, Jonker T, Offenbeek M, et al. Persisting workarounds in electronic health record system use: types, risks and benefits. BMC Med Inform Decis Mak. Published online June 8, 2021. https://doi.org/10.1186/s12911-021-01548-0
- Deng Y, Lin AC, Hingl J, et al. Risk factors for i.v. compounding errors when using an automated workflow management system. Am J Health Syst Pharm. 2016;73(12):887-893.
- Brasher C, Smith K, Daniels CC. Implementing an IV workflow management system. Pharmacy Purchasing & Products. 2022;19(4):2.
- Sexton JB, Thomas EJ, Helmreich RL, et al. High reliability organizations: a framework for improving patient safety in healthcare. Patient Saf Qual. 2014;18(1):1-10.
- King CA. To err is human, to drift is normalization of deviance. AORN J. 2010;91(2):284-286.
- ISMP. Maximize benefits of IV workflow management systems by addressing workarounds and errors. ISMP Medication Safety Alert! Acute Care. 2017;22(18). Accessed October 8, 2024. https://www.ismp.org/sites/default/files/attachments/2018-03/20170907.pdf
Copyright © 2024 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Addressing IV Workflow Management System Workarounds
Addressing IV Workflow Management System Workarounds