If you are under the impression that the long–awaited National Institute for Occupational Safety and Health (NIOSH) List of Hazardous Drugs in Healthcare Settings, 2024 is all you need to revamp your hazardous drug designations, there may be something you are missing!
The new NIOSH document states that it only contains new drug approvals or new safety-related warnings issued by the FDA from January 2014 through December 2015.1 As such, not all drugs entering the market Jan. 1, 2016 to the present have been reviewed for safety-related warnings issued by the FDA.
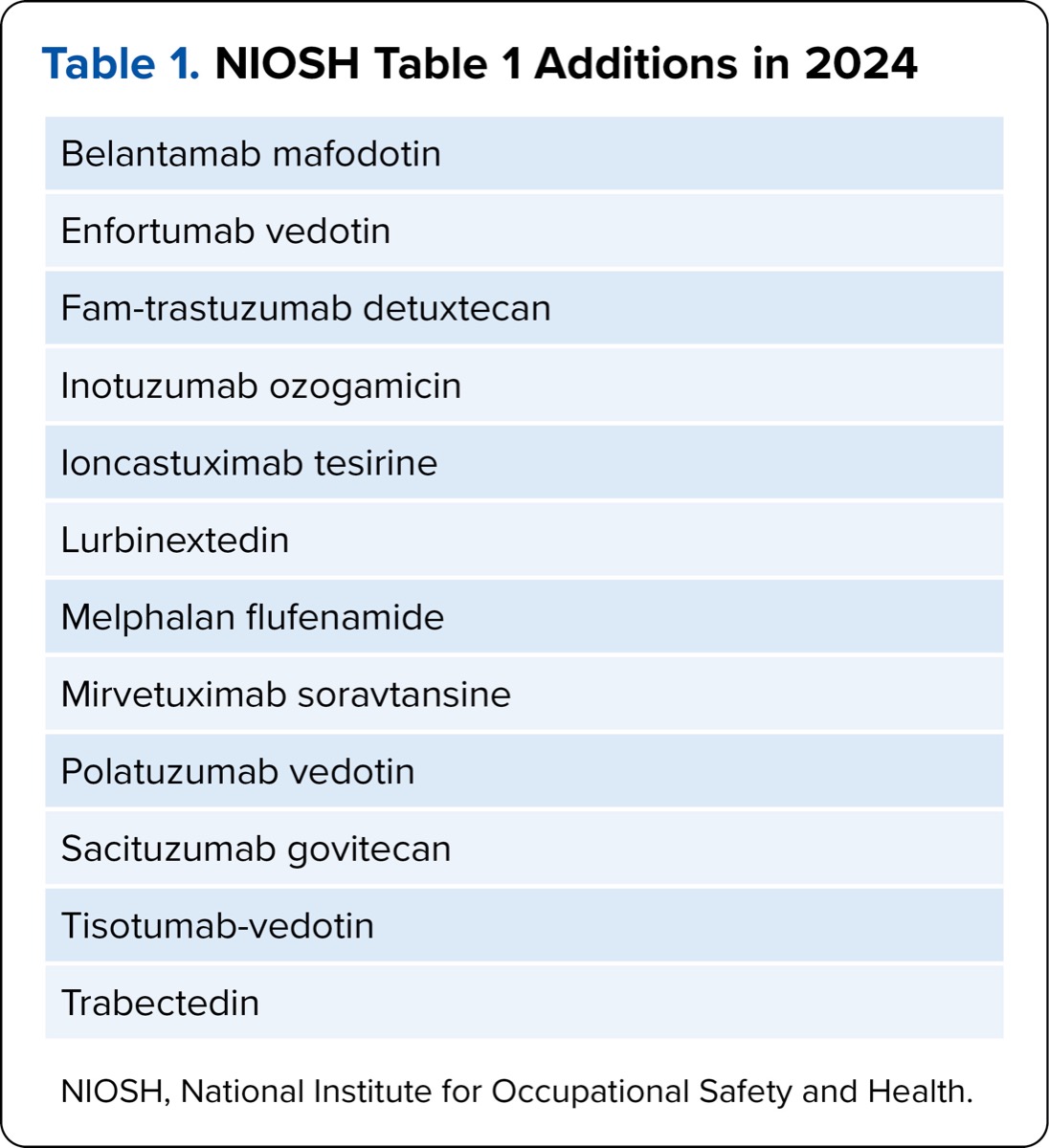
However, that’s not to say the NIOSH document is not helpful. For example, the authors have added 25 drugs to the 2024 list and removed 7 drugs from the 2016 list. These changes are posted to the NIOSH website,2 which supersedes the 2016 website. Moreover, the 2024 document has been streamlined, moving from 3 tables to 2. The definitions of drugs in the tables have also changed rather dramatically. The drugs listed in Table 1 now have precise definitions, which should prompt you to evaluate them with the utmost care. Not only do List 1 drugs now contain manufacturer’s special handling information (MSHI) in the package insert; they also meet 1 or more of the following criteria:
- classification as a known human carcinogen by the National Toxicology Program (NTP); and
- classification by the International Agency for Research on Cancer (IARC) as Group 1 “carcinogenic to humans” or Group 2A “probably carcinogenic to humans.”
| Table 1. NIOSH Table 1 Additions in 2024 |
| Belantamab mafodotin |
| Enfortumab vedotin |
| Fam-trastuzumab detuxtecan |
| Inotuzumab ozogamicin |
| Ioncastuximab tesirine |
| Lurbinextedin |
| Melphalan flufenamide |
| Mirvetuximab soravtansine |
| Polatuzumab vedotin |
| Sacituzumab govitecan |
| Tisotumab-vedotin |
| Trabectedin |
| NIOSH, National Institute for Occupational Safety and Health. |
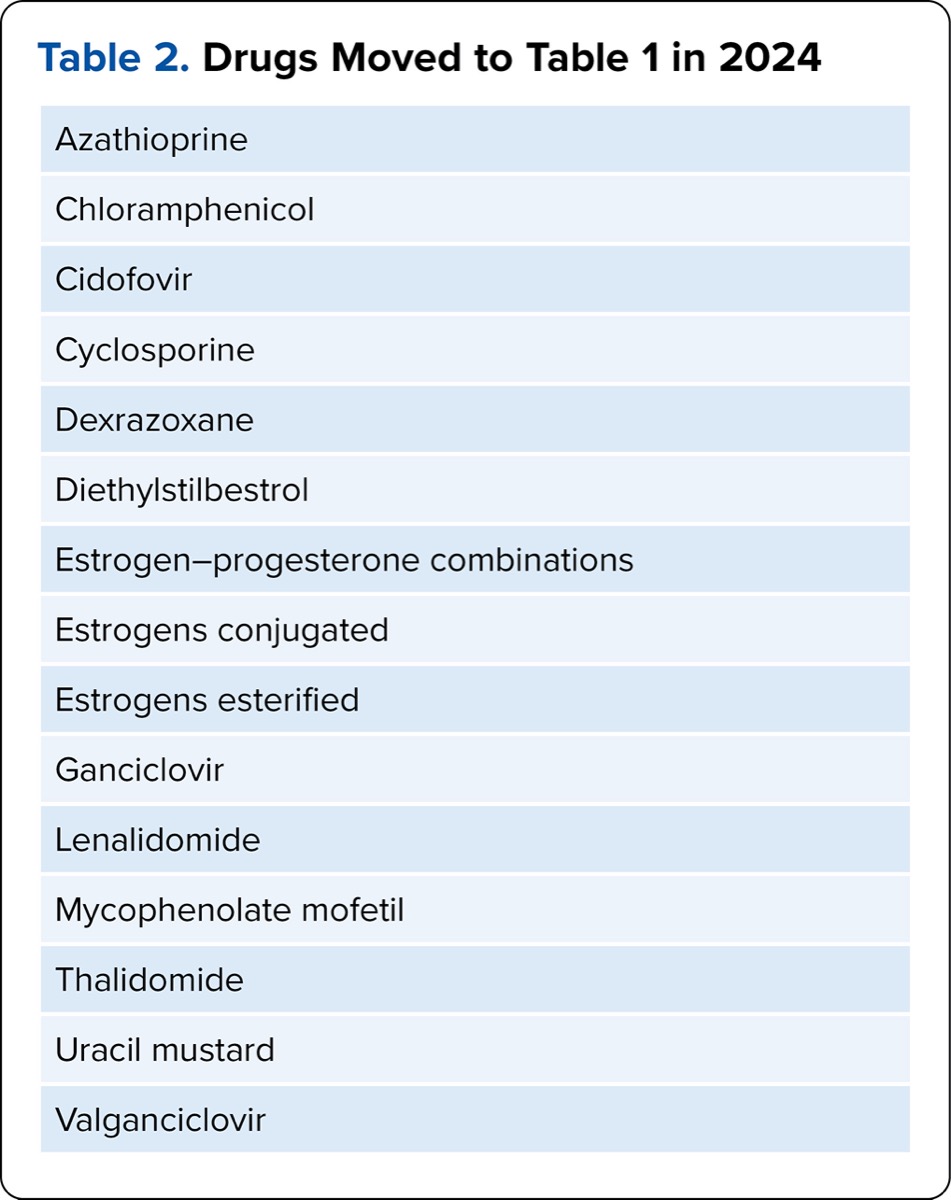
| Table 2. Drugs Moved to Table 1 in 2024 |
| Azathioprine |
| Chloramphenicol |
| Cidofovir |
| Cyclosporine |
| Dexrazoxane |
| Diethylstilbestrol |
| Estrogen–progesterone combinations |
| Estrogens conjugated |
| Estrogens esterified |
| Ganciclovir |
| Lenalidomide |
| Mycophenolate mofetil |
| Thalidomide |
| Uracil mustard |
| Valganciclovir |
These definitions also should apply to any new drugs entering the marketplace and your facility.
It is also important to note that not all of the drugs in the 2024 Table 1 list are antineoplastic drugs. That’s a big change from the 2016 list, which almost exclusively contained antineoplastics based on the AHFS classification. Additionally, only drugs approved by the FDA Center for Drug Evaluation and Research (CDER) are now considered for the NIOSH list. Drugs approved by the FDA Center for Biologics Evaluation and Research (CBER), including vaccines and biologicals, are not included in the 2024 NIOSH list. Some of these drugs, the most notable being Bacillus Calmette-Guérin (BCG), should also be included in the organization’s review of hazardous drugs.
So where does that leave the drugs in Table 2? These are drugs that do meet 1 or more of the criteria in the NIOSH definition of hazardous drug, which includes having adverse developmental and/or reproductive effects.
As for how organizations should use the 2024 NIOSH lists to update and maintain their USP General Chapter <800> hazardous drug list and related handling procedures, here are some important considerations. NIOSH has stated repeatedly that its role is to determine hazard identification, not to conduct a risk assessment. NIOSH does provide extensive guidance on how to conduct an assessment of risk in its earlier publication, Managing Hazardous Drug Exposures: Information for Healthcare Settings,3 published in April 2023. In this document, NIOSH specifically addresses the concepts of risk assessment, including the probability of occurrence and the severity of health effects.4 The Table of Control Approaches published in the 2023 document provides an excellent template for analyzing both the changes in the 2024 list and new hazardous drugs entering the marketplace.5
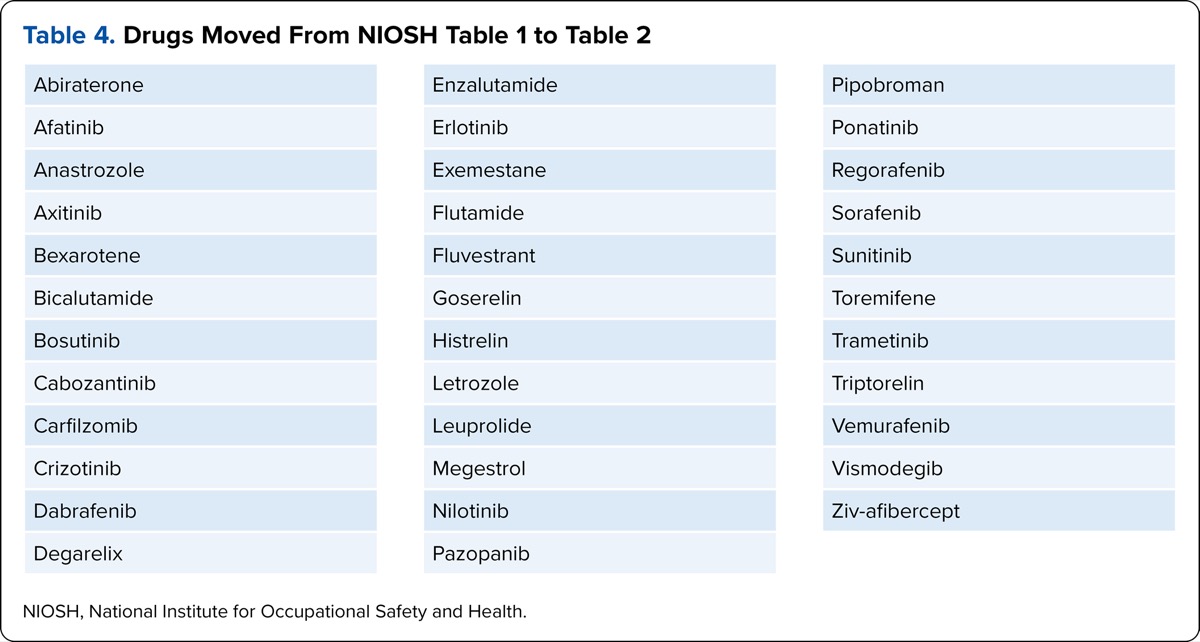
It will be important for organizations to consider a number of factors when making hazardous drug handling decisions based on the changes in the NIOSH criteria and the rapid evolution of drugs being approved by CBER. The first involves the definition of “antineoplastic.” Consider first the AHFS classification (AHFS 10.00), which is quite general in that it refers to drugs used in the treatment of cancer, some of which do not meet the NIOSH criteria for an antineoplastic hazardous drug. USP clarified the following in a memo effective July 1, 2020: “For the purposes of this chapter, the term antineoplastic only refers to antineoplastic drugs included in Table 1 of the most current NIOSH list.”6 This statement reduces the number of drugs considered to be active antineoplastics in Table 1 to 92, leaving 12 active non-antineoplastic drugs, with diethylstilbestrol and uracil mustard listed as legacy drugs no longer in use in the United States. Additionally, 35 hazardous drugs that did not have MSHI criteria were moved from Table 1 to Table 2.
| Table 3. NIOSH Table 2 Additions in 2024 |
| Blinatumomab |
| Ceritinib |
| Clobazam |
| Cobimetinib |
| Dihydroergotamine |
| Exenatide |
| Isotretinoin |
| Ivabradine |
| Lenvatinib |
| Miltefosine |
| Olaparib |
| Sonidegib |
| Urofollitropin |
| NIOSH, National Institute for Occupational Safety and Health. |
| Table 4. Drugs Moved From NIOSH Table 1 to Table 2 |
| Abiraterone |
| Afatinib |
| Anastrozole |
| Axitinib |
| Bexarotene |
| Bicalutamide |
| Bosutinib |
| Cabozantinib |
| Carfilzomib |
| Crizotinib |
| Dabrafenib |
| Degarelix |
| Enzalutamide |
| Erlotinib |
| Exemestane |
| Flutamide |
| Fluvestrant |
| Goserelin |
| Histrelin |
| Letrozole |
| Leuprolide |
| Megestrol |
| Nilotinib |
| Pazopanib |
| Pipobroman |
| Ponatinib |
| Regorafenib |
| Sorafenib |
| Sunitinib |
| Toremifene |
| Trametinib |
| Triptorelin |
| Vemurafenib |
| Vismodegib |
| Ziv-afibercept |
| NIOSH, National Institute for Occupational Safety and Health. |
One approach is to start from ground zero in the sense that the definitions of drugs in the list must be examined in terms of their relative risks. Second, drugs that are biologicals should be included by the organization for evaluation, perhaps using similar criteria as used by NIOSH for the CDER drugs. In addition to the example of BCG, chimeric antigen receptor (CAR) T-cell therapies, including anti-BCMA and anti-CD-19, will not be listed by NIOSH but should be evaluated.
Still, there is a large gray area of drugs that have entered the market after January 2016 and that meet the criteria for a hazardous drug but have not yet been evaluated by NIOSH. An interesting example is the recent addition, in November 2024, of hydrochlorothiazide, voriconazole, and tacrolimus to Group I of the IARC list of drugs classified as “carcinogenic to humans.”7 Although voriconazole and tacrolimus are already listed in NIOSH Table 2, hydrochlorothiazide is a new IARC addition. It should therefore be added to your organization’s list of hazardous drugs for consideration in terms of employee protection.
In a perfect world, all drugs used by your organization that are not on the NIOSH list would be reviewed using an available drug information system based on MSHI data, classification by the NTP as a known human carcinogen, or classified by IARC as a Group I human carcinogen.
Given time and staffing constraints, at least a review of the NTP and IARC lists would seem to be the most prudent approach. Ideally, every new drug being considered by the Pharmacy & Therapeutics (P&T) Committee should be scrutinized for hazardous drug characteristics and a decision made as to which Table NIOSH would logically assign it. Finally, once the drugs have been evaluated, risk decisions need to be made based on dosage forms and handling procedures, according to USP <800> standards. Hazardous drugs that are bulk active pharmaceutical ingredients and antineoplastic drugs in Table 1 requiring manipulation must be handled under full USP <800> containment requirements. Note again that the number of antineoplastic drugs in Table 1 of the new NIOSH list has been reduced, thereby allowing for more analysis and discretion. A hazardous drug risk assessment can be conducted for all other hazardous drugs to determine if the dosage form or route of administration can mitigate the need for respiratory controls, full personal protective equipment, and other protection practices. The particular dosage form and how it is being handled in certain situations, such as crushing of tablets by nurses, must all be considered. An excellent review of the criteria for drugs that are eligible for an assessment of risk, while slightly outdated in terms of the new NIOSH list, can be found in a USP FAQs document (bit.ly/3ETBsWp).8
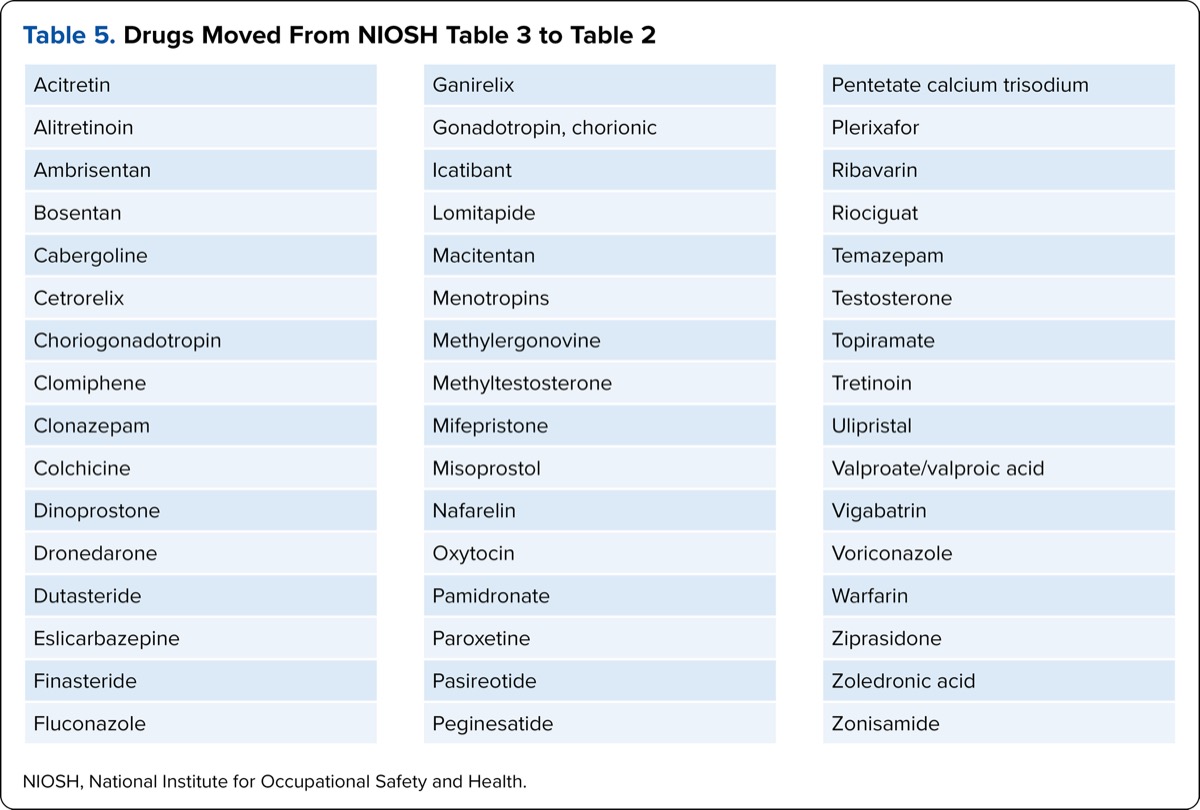
| Table 5. Drugs Moved From NIOSH Table 3 to Table 2 |
| Acitretin |
| Alitretinoin |
| Ambrisentan |
| Bosentan |
| Cabergoline |
| Cetrorelix |
| Choriogonadotropin |
| Clomiphene |
| Clonazepam |
| Colchicine |
| Dinoprostone |
| Dronedarone |
| Dutasteride |
| Eslicarbazepine |
| Finasteride |
| Fluconazole |
| Ganirelix |
| Gonadotropin, chorionic |
| Icatibant |
| Lomitapide |
| Macitentan |
| Menotropins |
| Methylergonovine |
| Methyltestosterone |
| Mifepristone |
| Misoprostol |
| Nafarelin |
| Oxytocin |
| Pamidronate |
| Paroxetine |
| Pasireotide |
| Peginesatide |
| Pentetate calcium trisodium |
| Plerixafor |
| Ribavarin |
| Riociguat |
| Temazepam |
| Testosterone |
| Topiramate |
| Tretinoin |
| Ulipristal |
| Valproate/valproic acid |
| Vigabatrin |
| Voriconazole |
| Warfarin |
| Ziprasidone |
| Zoledronic acid |
| Zonisamide |
| NIOSH, National Institute for Occupational Safety and Health. |
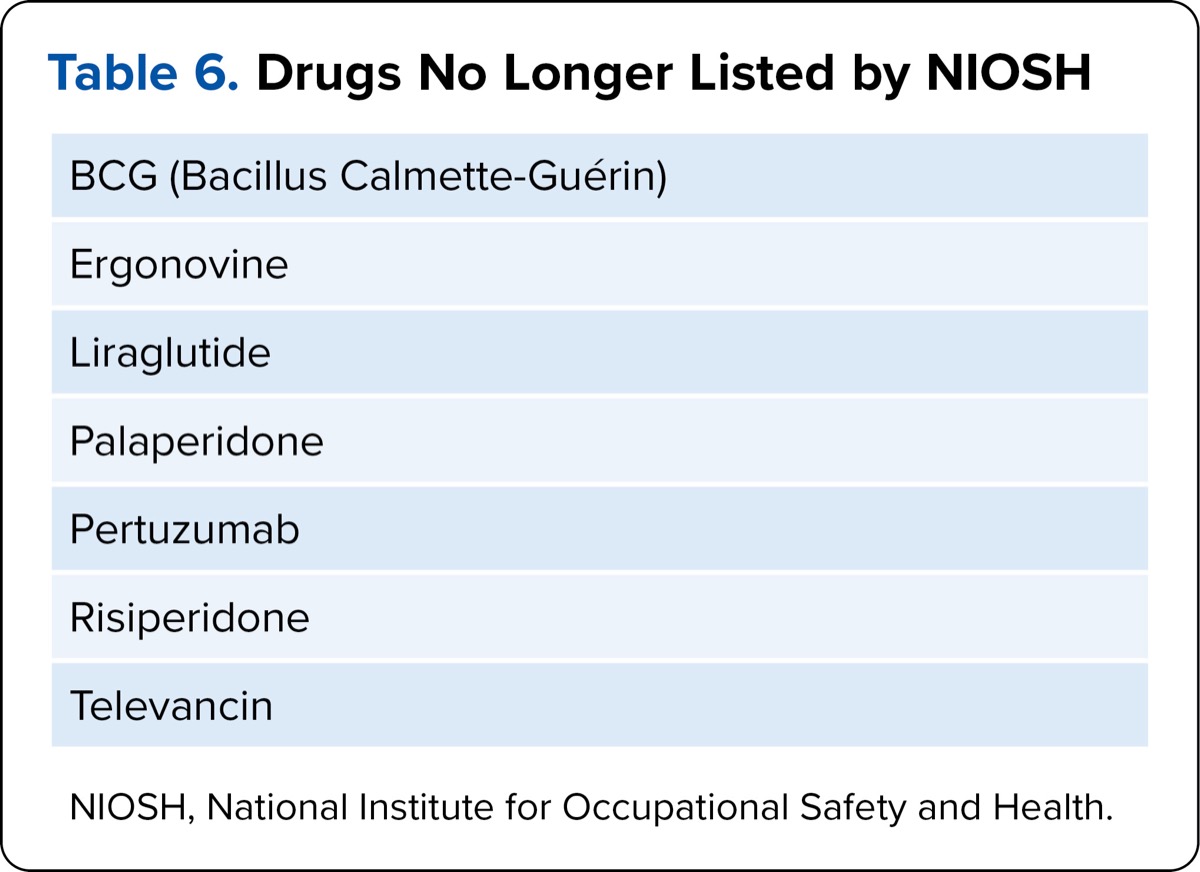
| Table 6. Drugs No Longer Listed by NIOSH |
| BCG (Bacillus Calmette-Guérin) |
| Ergonovine |
| Liraglutide |
| Palaperidone |
| Pertuzumab |
| Risiperidone |
| Televancin |
| NIOSH, National Institute for Occupational Safety and Health. |
To summarize the new changes, NIOSH moved a total of 35 drugs that appeared in 2016’s Table 1 to Table 2, and added 27 drugs for 2024. Those drugs added include immunosuppressive agents, chloramphenicol, anti-inflammatory agents, diethylstilbestrol, contraceptives, and estrogens. Seven drugs were removed completely from both lists: BCG, ergonovine, liraglutide, paliperidone, pertuzumab, risperidone, and telavancin. See our own Tables 1 to 6 for key changes to heed in NIOSH Tables 1 and 2.
An Opportunity for Improvement
These significant changes should stimulate your organization to review how it identifies and categorizes hazardous drugs and their handling. That review should focus on all employees involved in hazardous drugs handling , including pharmacy staff, nursing staff, surgical staff, radiology, and environmental services. Additionally, pharmacy purchasing professionals should be trained to flag new drugs for review, and any drugs being evaluated by the P&T Committee should include a hazard assessment and classification. State boards of pharmacy and the accrediting bodies will most certainly be assimilating these changes into their audits. If that isn’t enough incentive, your health and that of your colleagues should provide the motivation to get you started!
The authors reported no relevant financial disclosures beyond their stated employment.
References
- CDC. NIOSH List of Hazardous Drugs in Healthcare Settings, 2024. Published December 20, 2024. Accessed February 18, 2025. www.cdc.gov/niosh/docs/2025-103/default.html
- CDC. NIOSH (2023) Notice. Published May 9, 2023. Accessed February 18, 2025. www.cdc.gov/niosh/docs/2016-161/default.html
- CDC. Managing Hazardous Drug Exposures: Information for Healthcare Settings. Published April 2023. Accessed February 18, 2025. www.cdc.gov/niosh/docs/2023-130/default.html
- CDC. Managing Hazardous Drug Exposures: Information for Healthcare Settings. Published April 2023. Accessed February 18, 2025. www.cdc.gov/niosh/docs/2023-130/default.html (pp 6-7)
- CDC. Managing Hazardous Drug Exposures: Information for Healthcare Settings. Published April 2023. Accessed February 18, 2025. www.cdc.gov/niosh/docs/2023-130/default.html (pp 27-34)
- USP. <800> Hazardous Drugs—Handling in Healthcare Settings. Revision Bulletin. Published June 26, 2020. Accessed February 18. 2025. www.uspnf.com/sites/default/files/usp_pdf/EN/USPNF/revisions/gc-800-rb-notice-20200626.pdf
- IARC. Volume 137: Hydrochlorothiazide, voriconazole, and tacrolimus. Published November 29, 2024. Accessed February 18, 2025. https://monographs.iarc.who.int/news-events/volume-137-hydrochlorothiazide-voriconazole-and-tacrolimus/
- USP <800> FAQs. Updated November 21, 2023. Accessed February 18, 2025. bit.ly/3ETBsWp
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Does Your Hazardous Drug List Have Holes?
Does Your Hazardous Drug List Have Holes?