CSI Pharmacy
Texarkana, Texas
Immunoglobulin National Society
Calabasas, California
Immunoglobulin (Ig) therapy is an essential treatment for diverse immune-mediated and inflammatory disorders. Initially indicated for primary and secondary immunodeficiencies, its use has broadened to encompass immunomodulatory applications. Although clinically effective, intravenous immunoglobulin (IVIG) carries a risk for adverse drug reactions (ADRs) ranging from mild, transient symptoms to rare but severe complications.
Optimal IVIG administration requires coordinated efforts among nurses, pharmacists, and physicians. Key components include individualized risk assessment, informed product selection, adherence to standardized infusion protocols, and comprehensive patient education. Comprehensive pre-infusion evaluations, the use of validated patient-reported outcome measures (PROMs), and interdisciplinary collaboration support ongoing monitoring, guide therapeutic adjustments, and promote patient engagement.
This article presents a structured framework for safe and effective IVIG use, grounded in the Immunoglobulin National Society (IgNS) Standards of Practice (Edition 3.2). These evidence-based standards provide a legally defensible foundation for high-quality care, enhanced safety, and reduced complication rates. Specialized certification for Ig clinicians, Ig Certified Nurse (IgCN) and Ig Certified Pharmacist (IgCP), further reinforces clinical expertise and organizational quality.
Timely recognition, prevention, and management of ADRs are central to patient safety. Serious reactions such as thromboembolic events (TEEs), renal dysfunction, anaphylaxis, aseptic meningitis, and hemolysis require vigilant monitoring and evidence-based intervention strategies. Therapy optimization incorporates tailored dosing regimens, appropriate treatment intervals, IgG level monitoring where indicated, and tracking of disease-specific biomarkers.
By integrating risk mitigation strategies, interdisciplinary teamwork, infusion safety protocols, and shared decision-making, healthcare teams can maximize therapeutic outcomes while minimizing risks. The consistent application of the IgNS Standards of Practice, supported by clinician certification, safeguards patients, strengthens clinical practice, and elevates the overall quality of care.
Introduction
Ig therapy, administered intravenously, subcutaneously (SCIG), or as facilitated SCIG (fSCIG), has transformed the treatment landscape for numerous immunologic, autoimmune, and inflammatory disorders. Ig therapy exerts its action through two principal modalities:
- IgG replacement in patients with primary and secondary immunodeficiencies; and
- immunomodulation in those with autoimmune or inflammatory conditions such as chronic inflammatory demyelinating polyneuropathy (CIDP), multifocal motor neuropathy, dermatomyositis, and Kawasaki disease, among others.1-3
Ig therapy practice is a distinct and complex clinical specialty, requiring a nuanced understanding of pharmacologic principles and interdisciplinary coordination. As clinical use of Ig continues to expand, clinicians must understand the importance of patient-specific treatment protocols, including product selection, dosing, infusion rates, and treatment intervals, as well as metrics of treatment efficacy and disease progression. Clinicians must also perform extensive risk mitigation, appropriate assessments, and documentation required to maintain safety and manage ADRs. The IgNS Standards of Practice emphasizes the importance of individualized treatment planning and interdisciplinary care.2 Optimization begins with patient-specific assessment and product selection and continues through iterative care planning, consistent outcome evaluation, and effective team communication.
In patients with immunodeficiency diseases, although Ig therapy is used at lower doses, patient response must be carefully evaluated based on multiple factors, including serum IgG levels, incidence of infection, tolerability, clinical response, and others. More than 75% of IVIG therapy in the United States is used to treat autoimmune and inflammatory conditions, where higher doses and older patient populations are common, leading to greater risk and the need for a nuanced understanding of clinical context, patient characteristics, and therapy goals.4,5 For all patients receiving Ig therapy, clinical response, tolerability, and adherence are important outcome correlates.
The following sections focus on optimizing patient outcomes by examining the pathophysiology, risk factors, and clinical management of ADRs associated with IVIG therapy. Grounded in evidence-based recommendations from the IgNS Standards of Practice, this review provides clinicians with a structured, outcome-driven framework to enhance the safety, efficacy, and quality of Ig therapy across all care settings.
What Optimization Means in Ig Therapy
Optimization of IVIG therapy is not limited to the use of validated PROMs. Although PROMs provide important insight into functional status and patient experience, optimization must encompass a broader clinical framework that evaluates therapeutic efficacy, tolerability, safety, and alignment with patient goals.2,6-9
Monitoring therapy effectiveness
Optimization includes clinical and functional markers that track disease stability or progression. For primary immunodeficiency, this includes assessing for breakthrough infections, antibiotic use, and hospitalizations. In autoimmune or neurologic indications such as CIDP or myasthenia gravis, optimization includes monitoring for symptom exacerbation, relapses, or loss of treatment response. Clinicians may rely on serial assessments such as serum IgG levels (when appropriate), physical assessments and vital signs, validated scales and tools, and infection logs to determine whether the therapy remains effective.
Integrating PROMs as one part of a holistic strategy
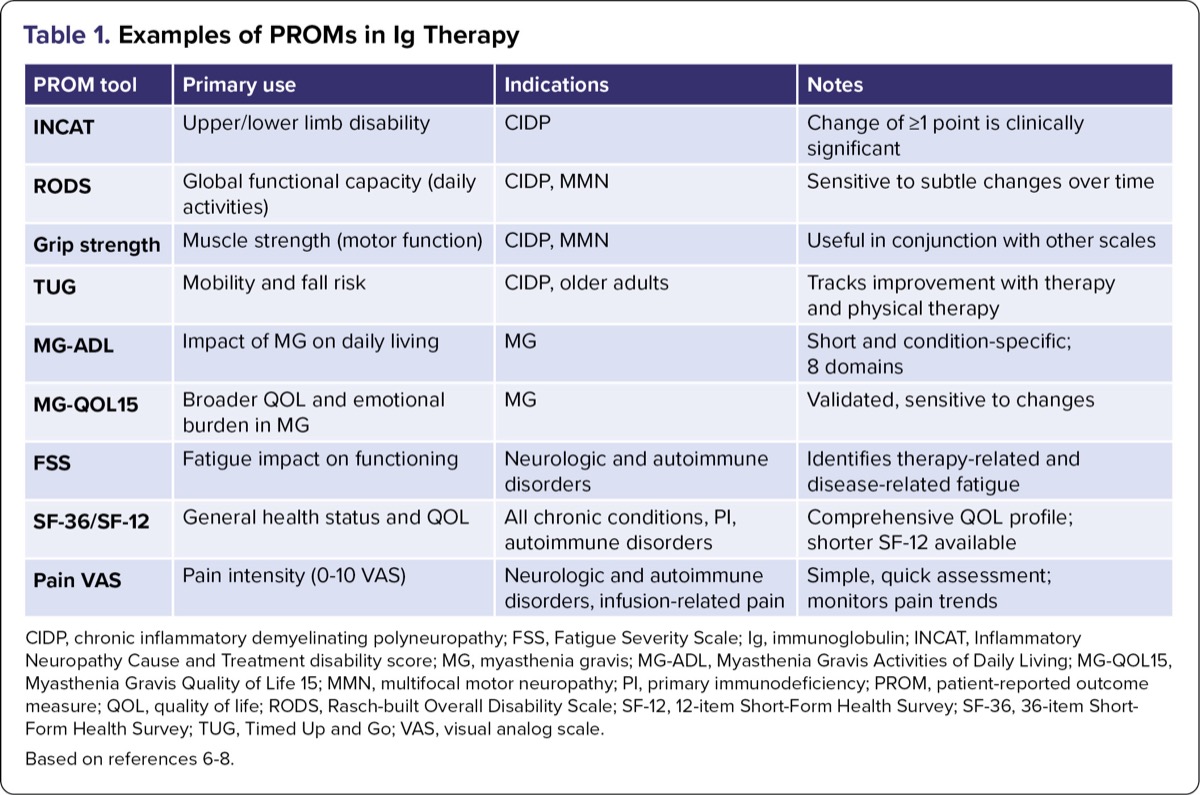
PROMs enhance clinical decision-making by capturing symptom and ADR severity, function, and satisfaction. However, they are not a replacement for clinical judgment, physical assessment, or laboratory monitoring. Used alongside clinician assessment, PROMs help support timely interventions and goal-oriented care (Table 1).
| Table 1. Examples of PROMs in Ig Therapy | |||
| PROM tool | Primary use | Indications | Notes |
|---|---|---|---|
| INCAT | Upper/lower limb disability | CIDP | Change of =1 point is clinically significant |
| RODS | Global functional capacity (daily activities) | CIDP, MMN | Sensitive to subtle changes over time |
| Grip strength | Muscle strength (motor function) | CIDP, MMN | Useful in conjunction with other scales |
| TUG | Mobility and fall risk | CIDP, older adults | Tracks improvement with therapy and physical therapy |
| MG-ADL | Impact of MG on daily living | MG | Short and condition-specific; 8 domains |
| MG-QOL15 | Broader QOL and emotional burden in MG | MG | Validated, sensitive to changes |
| FSS | Fatigue impact on functioning | Neurologic and autoimmune disorders | Identifies therapy-related and disease-related fatigue |
| SF-36/SF-12 | General health status and QOL | All chronic conditions, PI, autoimmune disorders | Comprehensive QOL profile; shorter SF-12 available |
| Pain VAS | Pain intensity (0-10 VAS) | Neurologic and autoimmune disorders, infusion-related pain | Simple, quick assessment; monitors pain trends |
| CIDP, chronic inflammatory demyelinating polyneuropathy; FSS, Fatigue Severity Scale; Ig, immunoglobulin; INCAT, Inflammatory Neuropathy Cause and Treatment disability score; MG, myasthenia gravis; MG-ADL, Myasthenia Gravis Activities of Daily Living; MG-QOL15, Myasthenia Gravis Quality of Life 15; MMN, multifocal motor neuropathy; PI, primary immunodeficiency; PROM, patient-reported outcome measure; QOL, quality of life; RODS, Rasch-built Overall Disability Scale; SF-12, 12-item Short-Form Health Survey; SF-36, 36-item Short-Form Health Survey; TUG, Timed Up and Go; VAS, visual analog scale. Based on references 6-8. | |||
Monitoring tolerability for patient safety
Ongoing evaluation for infusion-related ADRs is essential for optimizing Ig therapy treatment. This involves managing common systemic symptoms (eg, headache, hypotension, fatigue), identifying rare but serious adverse events (eg, thrombosis, hemolysis, aseptic meningitis), and adjusting therapy as needed. Rate titration, premedication, hydration strategies, and changes in brand or route may be necessary to maintain tolerability.
Incorporating patient-defined goals
Treatment optimization must reflect what matters most to the patient. This includes the preferred route of administration (IVIG vs SCIG vs fSCIG), site of care (home, infusion center, clinic), frequency of infusions, and level of autonomy (self-administration vs nurse administered). These choices directly affect adherence and quality of life. Shared decision-making is essential for aligning therapy with a patient's life phase, lifestyle, work or school obligations, and caregiver needs.
Facilitating interdisciplinary and iterative care
Optimization is not static. It requires continuous collaboration between pharmacists, nurses, and physicians to assess risks, modify care plans, and respond to evolving needs. Therapy may need to be reevaluated and adjusted based on seasonal infections, comorbid conditions, or patient-reported experiences.
General Prevention Strategies For Managing ADRs
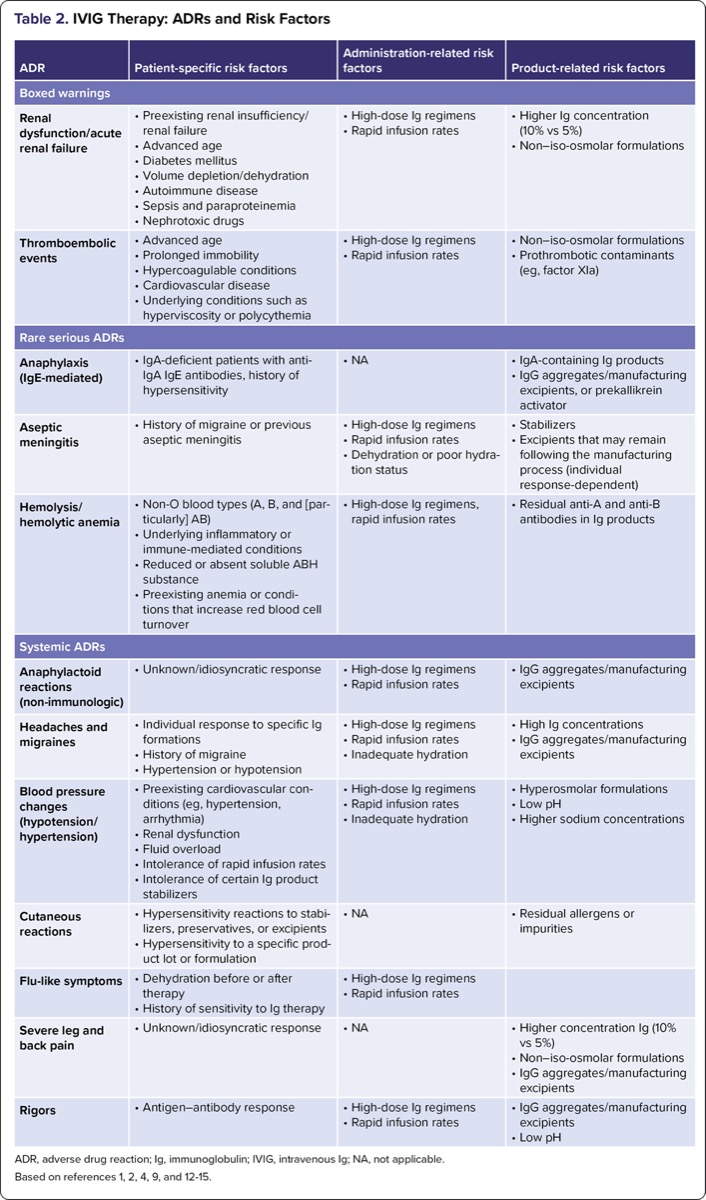
Many ADRs associated with IVIG therapy share overlapping risk factors and corresponding mitigation strategies, which are labeled as patient-specific, administration-related, or product-specific categories (Tables 2 and 3). Implementing proactive, general prevention measures across all patients can significantly reduce both the incidence and severity of these complications.2,9
- Pre-infusion assessment is an essential foundation for safe infusion. It should include a thorough review of the patient’s history, recent changes in health status, current prescription and over-the-counter medications and supplements, pertinent laboratory values, baseline vital signs, and hydration status.
- Hydration plays a central role in minimizing ADRs. Patients should be encouraged to maintain adequate hydration before, during, and after the infusion. For those at higher risk for ADRs, such as older adults or patients receiving high doses of Ig, IV fluids may be necessary, unless a fluid restriction is present. Proper hydration can help reduce the risk for common ADRs such as headaches and flu-like symptoms and serious adverse events such as aseptic meningitis, TEEs, and renal dysfunction.
- Product selection should be individualized and based on product risk factors (Table 2). Factors such as physiologic osmolality, product stabilizers, and impurities and excipients may affect tolerability. In patients at risk for hemolysis, and in those with blood types other than O, formulations with lower anti-A and anti-B titers should be chosen. Patients with diabetes should avoid glucose-stabilized products. Patients with renal dysfunction should avoid products containing sodium. In patients with recurrent tolerability issues that persist despite risk mitigation efforts, switching to an alternate IVIG brand or transitioning to SCIG may be considered.
- Upon starting the infusion, in the event of an ADR, immediate assessment of the patient is critical to determine the severity and gauge the progression of the reaction. Clinicians must be prepared to pause or discontinue the infusion as necessary, document clinical findings, and promptly communicate with the prescriber when symptoms do not resolve with standard interventions or when signs of escalation are observed. Early recognition and timely collaboration are essential to prevent the progression of reaction severity and ensure the appropriate management of more serious adverse events.
- IVIG therapy requires specific infusion protocols that involve a careful and multistep titration schedule. Infusions should start at the slowest rate practicable (determined by many factors, including total dose, weight, patient risk factors, and any prior infusion-related issues) with gradual titration based on patient tolerability. When feasible, consider splitting doses (especially doses =1-2 g/kg) over consecutive or alternate (with prescriber order) days to reduce the risk for adverse reactions.
- Premedication with antihistamines and analgesics may be appropriate for patients at an increased risk for adverse reactions to Ig therapy under physician orders. (Corticosteroids may also be used in high-risk individuals.) These include individuals with a history of infusion-related symptoms, documented hypersensitivities, or recurrent migraines. Premedication can reduce the incidence and severity of systemic reactions by mitigating inflammatory and immune-mediated responses.
| Table 2. IVIG Therapy: ADRs and Risk Factors | |||
| ADR | Patient-specific risk factors | Administration-related risk factors | Product-related risk factors |
|---|---|---|---|
| Boxed warnings | |||
| Renal dysfunction/acute renal failure |
|
|
|
| Thromboembolic events |
|
|
|
| Rare serious ADRs | |||
| Anaphylaxis (IgE-mediated) |
|
|
|
| Aseptic meningitis |
|
|
|
| Hemolysis/ hemolytic anemia |
|
|
|
| Systemic ADRs | |||
| Anaphylactoid reactions (non-immunologic) |
|
|
|
| Headaches and migraines |
|
|
|
| Blood pressure changes (hypotension/hypertension) |
|
|
|
| Cutaneous reactions |
|
|
|
| Flu-like symptoms |
|
| |
| Severe leg and back pain |
|
|
|
| Rigors |
|
|
|
| ADR, adverse drug reaction; Ig, immunoglobulin; IVIG, intravenous Ig; NA, not applicable. Based on references 1, 2, 4, 9, and 12-15. | |||
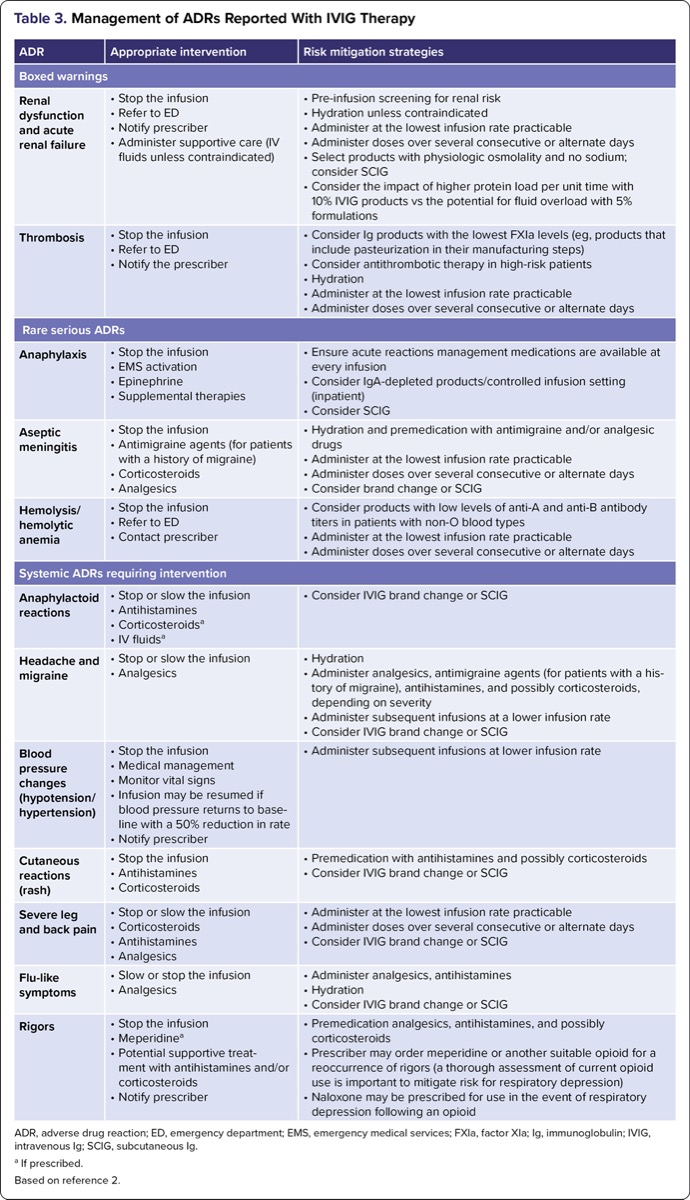
| Table 3. Management of ADRs Reported With IVIG Therapy | ||
| ADR | Appropriate intervention | Risk mitigation strategies |
|---|---|---|
| Boxed warnings | ||
| Renal dysfunction and acute renal failure |
|
|
| Thrombosis |
|
|
| Rare serious ADRs | ||
| Anaphylaxis |
|
|
| Aseptic meningitis |
|
|
| Hemolysis/hemolytic anemia |
|
|
| Systemic ADRs requiring intervention | ||
| Anaphylactoid reactions |
|
|
| Headache and migraine |
|
|
| Blood pressure changes (hypotension/hypertension) |
|
|
| Cutaneous reactions (rash) |
|
|
| Severe leg and back pain |
|
|
| Flu-like symptoms |
|
|
| Rigors |
|
|
| ADR, adverse drug reaction; ED, emergency department; EMS, emergency medical services; FXIa, factor XIa; Ig, immunoglobulin; IVIG, intravenous Ig; SCIG, subcutaneous Ig. a If prescribed. Based on reference 2. | ||
Clinicians should follow evidence-based standards for premedication in patients at risk for infusion-related adverse events, as outlined in the IgNS Standards of Practice and product-specific guidance. In addition, clinicians should stay informed of emerging safety trends by monitoring public health surveillance systems such as the FDA’s MedWatch and the CDC initiatives, which support the ongoing evaluation and regulation of blood-derived therapeutics.2,10,11
Optimizing Ig Therapy: Use of Validated PROMs
With Ig therapy primarily administered in the home and outpatient infusion settings, the use of validated PROMs remains a critical component of patient care, assessing clinical effectiveness and guiding iterative care (Table 1). PROMs offer valuable insight into the patient’s perspective on treatment impact, particularly in areas such as fatigue, muscle strength, mobility, and activities of daily living.6-8
PROMs not only capture the patient’s functional status and symptom trends but also provide critical insight into health-related quality of life, including the psychological, social, and physical burden of disease and treatment. These patient-centered tools provide standardized, reproducible data that complement clinician observations and support earlier recognition of treatment decline or adverse events. By embedding PROMs into routine care, clinicians can better understand the evolving impact of therapy on patients’ daily lives, enhance engagement, and guide timely, goal-aligned adjustments to care plans. PROMs are simple to administer and can be used across various care settings, including infusion clinics, neurology practices, and home health, to help assess responses and adjust therapy accordingly.6-8
Clinical Considerations2
The healthcare team plays a vital role in enhancing patient safety with IVIG, from administration to outcomes. Key responsibilities include patient-specific risk mitigation, assessment, monitoring, documentation, and appropriate interventions. Through comprehensive evaluation and ongoing communication with patients, pharmacists, nurses, and physicians, the healthcare team ensures early detection and effective management of therapy-related complications.
Before initiating the infusion, the pharmacist and nurse should perform a comprehensive patient assessment to identify potential risk factors (Table 2). This assessment includes a review of the patient’s medical history for prior infusion reactions, hypersensitivities, and comorbidities such as migraine, renal impairment, or autoimmune conditions. It is also essential to evaluate for the presence of infection by assessing for elevated temperature or signs and symptoms of illness, including sepsis, such as chills, tachycardia, hypotension, or altered mental status. Baseline vital signs, hydration status, and pertinent laboratory values, including renal function, hemoglobin levels, and blood type, should be obtained and documented. Nurses must verify that any prescribed drugs and premedication have been administered and confirm that the infusion setting is adequately equipped with acute reaction management needs—such as an anaphylaxis kit—in the event of an ADR. Pharmacists and nurses should manage ADRs with specialized skills and competencies in Ig therapy, consulting with the physician and adhering to best clinical practice and organizational protocols.
Throughout the infusion process, clinicians should monitor for mild, moderate, severe, and rare serious reactions. Vital signs should be assessed at regular intervals per the IgNS Standards of Practice and organizational protocols. Additionally, clinicians must promptly recognize early-warning signs such as rash, chest tightness, dizziness, hypotension, headache, dyspnea, or changes in mental status, and be prepared to stop the infusion and initiate prescriber notification or emergency protocols when needed.
After the infusion, clinicians should continue monitoring patients for delayed reactions, such as signs of hemolysis or aseptic meningitis. They should reinforce patient education, emphasizing the importance of reporting symptoms of serious adverse events, including but not limited to dark urine, severe headache, shortness of breath, and swelling. Thorough documentation of infusion details, patient response, and interventions ensures continuity of care and supports pharmacovigilance.
Clinicians affect treatment success by educating and empowering patients to understand their treatment goals, risks, and strategies to improve the infusion experience, including hydration, monitoring, communication, and follow-up. Nurses’ competence and clinical judgment are essential to the safe and effective administration of Ig therapy across all diagnoses, care settings, and routes of administration.
Clinical Management of Rare Serious Adverse Events: Boxed Warnings, Contraindications, and Precautions
For more information, refer to the IgNS Standards of Practice and Table 2, which outlines risk factors associated with IVIG therapy.2
Renal dysfunction and acute renal failure
Renal dysfunction and acute renal failure are rare but serious complications of IVIG therapy. Historically, sucrose-stabilized IVIG products were implicated in the disproportionate number of renal ADRs. However, at the time of publication, no FDA-approved Ig therapy brands are stabilized with sucrose.4
The risk for renal dysfunction has been attributed to a combination of product- and patient-related factors, as outlined in Table 2.4
Pathophysiology of renal dysfunction
Several mechanisms contribute to IVIG-associated renal injury4,16:
- High plasma viscosity can reduce renal blood flow and filtration.
- Metabolic burden from stabilizers and aggregates increases renal workload.
- Tubular obstruction or toxicity may result from Ig aggregates or residual proteins.
- Complement-mediated inflammation may trigger or worsen renal injury.
Mitigation and management (pharmacy and nursing)2,4
- Risk mitigation begins with screening for known risk factors (Table 2) and review of baseline renal laboratory tests, including blood urea nitrogen, serum creatinine, and estimated glomerular filtration rate. These values should be monitored regularly, and adjustments made if renal function declines.
- Product selection should favor IVIG formulations with physiologic osmolality and low sodium. For high-risk patients, 5% products may be preferred to reduce protein load; however, they carry an increased risk for fluid overload. Conversely, formulations with 10% protein concentration should be used with caution due to the potential for an increased renal burden.
- Hydration is crucial for minimizing renal strain. Encourage oral fluids or administer IV hydration before infusion, unless contraindicated.
- Infusion protocols should prioritize the slowest initial rates practicable, with doses administered over multiple consecutive or alternate days. Discontinue IVIG if renal function deteriorates despite precautions.
Clinical considerations2
Clinicians should monitor for signs of renal dysfunction, including renal laboratory values. If signs and symptoms of renal impairment occur, such as decreased urine output, sudden weight gain, fluid retention, swelling in the lower extremities, back or flank pain, or changes in blood pressure, stop the infusion immediately, assess the patient, notify the prescriber, activate 911/emergency medical services if patient is in acute distress, and initiate appropriate supportive measures. Early recognition and intervention are essential to prevent progression to acute renal failure, particularly in high-risk patients receiving IVIG therapy. For patients with ongoing renal concerns or prior complications with IVIG, consider transitioning to SCIG.
Thromboembolic events
TEEs are rare but serious complications of IVIG therapy and can result in deep vein thrombosis, pulmonary embolism, stroke, or myocardial infarction. These events arise from both patient-specific risk factors and product-related characteristics that promote hypercoagulability and impaired circulation (Table 2).17,18
Pathophysiology
Multiple mechanisms may contribute to IVIG-associated thrombosis2,15,17,19:
- Plasma viscosity. Risk factors include advanced age, prolonged immobilization, hypercoagulable conditions, and a history of thrombosis. Rapid increases in serum IgG can elevate plasma viscosity, thereby reducing microvascular flow and potentially promoting clot formation.
- Platelet aggregation. High IgG concentrations may directly or indirectly enhance platelet activation.
- Prothrombotic contaminants. Factors such as prekallikrein activator (PKA) and factor XIa (FXIa) may activate clotting pathways.
- Erythrocyte and leukocyte aggregation. Aggregation of red and white blood cells may further obstruct microcirculation.
- Vasospasm and cytokine release. IVIG may trigger cytokine release and arterial vasospasm, leading to endothelial damage and an increased risk of clotting.
- Hyperosmolarity. IVIG products with high osmolality may increase vascular resistance, especially in high-risk individuals with advanced age and underlying conditions such as hyperviscosity or polycythemia.
Mitigation and management (pharmacy and nursing)2,15
- Before infusion, assess for cardiovascular disease, immobility, history of thrombosis, or hypercoagulable conditions. For high-risk patients, obtain baseline coagulation laboratory markers such as prothrombin time, activated partial thromboplastin time, and international normalized ratio as appropriate.
- Ensure patients are well hydrated before, during, and after infusion to reduce plasma viscosity. Use IV fluids when needed unless contraindicated.
- Initiate IVIG at the lowest infusion rate practicable, and administer doses over multiple consecutive or alternate days. For high-risk patients, discuss prophylactic measures (such as antithrombotic agents) with the prescriber.
Product considerations
When selecting an IVIG product, evaluate its manufacturing process and composition, including stabilizers. Techniques such as affinity chromatography and pasteurization help remove prothrombotic proteins, including FXIa and PKA.2
For patients with a history of thrombosis, SCIG may offer a safer alternative.2,9
Clinician considerations2,15
- Monitor closely throughout the entire infusion.
- Before infusion, nurses should conduct a comprehensive assessment, including reviewing the patient’s history, prescribed and over-the-counter medications, hydration status, and mobility impairments. Educate the patient on signs and symptoms of TEEs, such as chest pain; shortness of breath; swelling, warmth, redness, or discoloration of the leg; pain or tenderness; or sudden weakness.
- Stop the infusion immediately and notify the prescriber if symptoms such as chest pain, shortness of breath, numbness, neurologic changes, or unilateral swelling appear.
- After the infusion, monitor for delayed symptoms, such as shortness of breath or leg pain, particularly in high-risk patients. Reinforce mobility, hydration, and follow-up care.
- All observations, interventions, and patient-reported symptoms should be thoroughly documented.
Anaphylaxis: recognition, risk, and management
Anaphylactic or severe hypersensitivity reactions to Ig products are rare but serious and are listed as a class-labeled contraindication.1,2,12
Pathophysiology and clinical presentation
True anaphylaxis to Ig therapy involves an IgE-mediated cascade involving mast cell and basophil activation with release of histamine and other mediators. Triggering factors may additionally include IgG aggregates, fragments, or PKA in the product.1,2,12 Symptoms can develop rapidly and include severe hypotension, respiratory distress, and in some cases, cardiopulmonary arrest.2
Mitigation and management (pharmacy and nursing)1,2,12
- Immediate recognition and emergency intervention are critical to patient survival.
- Patients at greatest risk include those with selective IgA deficiency (<1.2 mg/dL) and anti-IgA antibodies of the IgE type, and a history of hypersensitivity to blood products.
- Of note, IgG anti-IgA antibodies are not strongly associated with severe reactions. Some patients with anti-IgA reactions may tolerate low-IgA Ig therapy formulations (0.4-2.9 mcg/mL).
Emergency preparedness and management2,12
In high-risk patients, infusions should occur in controlled clinical environments equipped with the ability to manage acute reactions. These should include, at a minimum, epinephrine and antihistamines, and in some instances IV fluids, corticosteroids, and a bronchodilator under a physician’s orders. The infusion should always begin at the slowest practicable rate, with gradual titration based on patient tolerance. Patients should receive premedication with antihistamines and corticosteroids. IgA-depleted Ig products can help reduce risk.
Clinician considerations2,12
Before infusion, the clinician should review the patient’s medical history, confirm premedication, and ensure emergency supplies are readily available. Continuous monitoring of vital signs is essential before, during, and after infusion in a setting equipped to manage acute reactions.
Aseptic meningitis syndrome (AMS)
AMS typically presents as a severe, persistent headache, often accompanied by nausea, neck stiffness, and light sensitivity, and can last from several hours to a few days. Although the condition is self-limiting, it can be debilitating and distressing to patients and often requires clinical interventions.1,2,20
Pathophysiology and risk factors
Several mechanisms have been proposed to explain AMS. One is cytokine-mediated inflammation triggered by the Ig infusion and osmotic shifts. Other mechanisms include hypersensitivity reactions to stabilizers or excipients, localized inflammation from Ig crossing the blood–brain barrier, and neutrophilic pleocytosis in the cerebrospinal fluid.1,20,21
Mitigation and management (pharmacy and nursing)2
- Before initiating IVIG, assess for a history of migraines or past episodes of AMS. Tailor the treatment plan accordingly, especially for high-risk patients.2
- Ensure the patient is well hydrated before, during, and after the infusion. If needed, administer IV fluids unless contraindicated. Infuse at the slowest rate practicable and increase only if well tolerated. Administer doses over several consecutive or alternate days. Consider switching to a different IVIG brand. For recurrent AMS, transitioning to SCIG may help, as it avoids rapid IgG serum peaks associated with IVIG.2
- Because AMS symptoms mimic those of infectious meningitis, a thorough evaluation is required to rule out infection and confirm diagnosis.2
- For symptom relief, consider antimigraine agents such as triptans, beta-blockers, or calcium channel blockers, especially in patients with a known migraine history. Anti-inflammatory options, such as IV corticosteroids, may help reduce inflammation and discomfort. Tailor pain management to the individual patient’s needs.2
Clinical considerations
Nurses should identify at-risk patients early, monitor for symptoms during and after the infusion, educate patients on what to expect and when to report symptoms, and document and communicate all symptoms and interventions clearly.2
Hemolysis and hemolytic anemia
Hemolysis, or the destruction of red blood cells, is a rare but potentially serious complication of IVIG therapy. In some cases, hemolysis can lead to hemolytic anemia, and in severe instances, may contribute to renal dysfunction or disseminated intravascular coagulation.2,13,22
Pathophysiology
Hemolysis is primarily caused by the passive transfer of anti-A and anti-B isoagglutinins from IVIG products derived from pooled plasma. These antibodies can bind to red blood cells in patients with non-O blood types (A, B, or AB), triggering immune-mediated destruction via complement activation, antibody-dependent cellular cytotoxicity, or enhanced splenic clearance.13,22
Clinical presentation
Hemolytic anemia may present 2 to 10 days post-infusion, with symptoms such as2,13:
- fatigue, pallor, shortness of breath, and tachycardia;
- jaundice (from elevated bilirubin); and
- dark urine (due to hemoglobinuria).
- Laboratory findings typically include13:
- low hemoglobin and hematocrit;
- elevated lactate dehydrogenase (LDH) and indirect bilirubin;
- low haptoglobin; and
- a positive direct antiglobulin test for IgG or C3.
Mitigation and management (pharmacy and nursing)2
- Prior to infusion, assess for hemolysis risk: blood type, anemia history, autoimmune disease, and baseline labs tests (hemoglobin, hematocrit, LDH, bilirubin, haptoglobin).
- Ensure adequate fluid intake before, during, and after infusion. Use IV fluids if oral hydration is insufficient and not contraindicated.
- Use the lowest feasible rate and titrate cautiously. Divide doses over multiple consecutive or alternate days.
- Select formulations with low anti-A and anti-B titers when available. Consider SCIG for high-risk patients or those with prior reactions.
Clinical management of hemolysis based on severity2,13
- Mild: supportive care and close monitoring
- Moderate: IV fluids, folic acid, and erythropoiesis support
- Severe: red blood cell transfusion; corticosteroids for immune-mediated cases
Clinical management of other systemic ADRs
Systemic ADRs such as anaphylactoid reactions, headache, blood pressure fluctuations, cutaneous reactions, flu-like symptoms, severe leg and back pain, and rigors and may be attributed to complement activation, cytokine release, or vasoactive mediator release.14,23 Such ADRs can often be managed by administering Ig therapy at the slowest infusion rate practicable. Prompt recognition and supportive management are key to ensuring patient safety and comfort (Tables 2 and 3).
For more information, refer to the IgNS Standards of Practice and Table 2 outlining risk factors associated with IVIG therapy.2
Anaphylactoid reactions (non-immunologic)1,2,14,23
Anaphylactoid (also known as non-immunologic) reactions may resemble anaphylaxis but are not mediated by anti-IgA IgE antibodies, are slower to onset, and less severe than anaphylaxis. Anaphylactoid reactions may be triggered by cytokine release, IgG aggregates, or excipients in the product. Symptoms may include flushing, back pain, nausea, hypertension, or chest tightness.
Mitigation and management (pharmacy and nursing)2
- Premedication with antihistamines and corticosteroids may help reduce the likelihood in at-risk individuals.
- Slow infusion rates are essential.
- If symptoms occur, pause the infusion, monitor vitals, and provide supportive care.
- Restart cautiously only if symptoms resolve and under prescriber guidance.
Headache
One of the most common Ig therapy ADRs, headache may occur because of a combination of factors, including meningeal irritation, cytokine-mediated inflammation, and rapid elevations in serum IgG levels, particularly with high doses or high infusion rates. Additional contributing factors may include fluctuations in blood pressure, dehydration, nonphysiologic osmolality, or excipient profile of the Ig product (Table 2).20,23,24
Mitigation and management (pharmacy and nursing)1,2
- Ensure adequate hydration before and after infusion.
- Premedicate with analgesics or antimigraine agents in patients with a known history of migraine.
- Reduce infusion rate and consider administering doses over multiple consecutive or alternate days.
- In recurrent cases, transitioning to SCIG may be helpful.
Blood pressure changes
Blood pressure fluctuations during IVIG therapy are not uncommon and may present as either hypotension or hypertension, depending on patient-specific factors and the infusion context (Table 2). Hypotension may result from fluid shifts associated with the osmolality of the IVIG product, intravascular volume depletion, or vasodilatory responses triggered by cytokine release. In some cases, hypersensitivity-type reactions can also cause transient vasodilation and hypotension. Conversely, hypertension may occur in patients with preexisting autonomic dysfunction, such as those with neurologic conditions, or may be linked to infusion-related anxiety, pain, or discomfort. Additionally, rapid infusion rates can lead to sympathetic nervous system stimulation, contributing to elevated blood pressure during or after the infusion.1,2
Mitigation and management (pharmacy and nursing)1,2
- Monitor blood pressure closely throughout the infusion.
- Hypotension: Pause infusion, position patient flat, and provide fluids.
- Hypertension: Pause or reduce the rate by 50%, continue to monitor patient blood pressure, and notify the prescriber.
- Cutaneous reactions:
- Mild urticaria, rash, or flushing may occur, especially early during IVIG infusion.1,2
Mitigation and management (pharmacy and nursing)2
- Treat mild symptoms with oral antihistamines.
- Reduce the infusion rate.
- For persistent or worsening reactions, contact the prescriber and activate emergency medical services for acute reactions involving airway/breathing compromise.
- Consider corticosteroids or switching the Ig product.
Discussion
IVIG therapy remains the gold standard in the treatment of immunologic, autoimmune, and neuroinflammatory disorders. Although generally well tolerated, IVIG administration carries the risk for ADRs, ranging from mild infusion-related symptoms to rare but serious complications, including TEEs, anaphylaxis, and hemolysis. Timely recognition and management of these events are essential to reducing morbidity and ensuring patient safety, as emphasized in the IgNS Standards of Practice.2
The prevention and management of IVIG-related ADRs require a coordinated, interdisciplinary approach. Nurses, pharmacists, and physicians play a critical role in individualized treatment planning, product selection, risk assessment, infusion safety, and patient education. Shared decision-making with the patient enhances adherence and promotes individualized care. Pharmacists should evaluate preexisting patient risk factors and product-specific properties that can affect safety. Nurses should monitor tolerability, implement safety protocols, and respond to infusion-related events in real time.2
Optimization of IVIG therapy extends beyond the prevention of adverse events. It includes careful consideration of the product formulation, route of administration, and infusion schedule to align therapy with the patient’s clinical profile, response, tolerability, and personal preferences. PROMs such as validated functional scales play an increasingly important role in guiding iterative care planning and assessing the efficacy of IVIG therapy over time.
Documentation is essential to every infusion. Consistent and thorough records of pre-infusion assessments, adverse events, interventions, and outcomes support care continuity and regulatory compliance. Severe and serious ADRs must be reported through appropriate channels (FDA MedWatch) to support broader pharmacovigilance.2,11
The introduction of new Ig formulations, shifts in product availability, and the emergence of updated safety data necessitate ongoing clinician engagement with current evidence and guidelines. While external resources such as updates from the IgNS, the CDC, and the FDA provide critical guidance, internal mechanisms, including case-based learning, adverse event debriefings, and interdisciplinary reviews, serve as valuable tools for reinforcing best practices and fostering a culture of clinical vigilance and shared accountability.2,10,25
Despite improvements in clinician education and pharmacovigilance, substantial gaps remain in the evidence surrounding the incidence, mechanisms, and management of IVIG-related ADRs. Future research should focus on validating screening tools, comparing risk across products, and developing patient-facing technologies to improve real-time ADR reporting. Investing in interdisciplinary training and patient-centered care models remains essential to improve outcomes and reduce preventable complications in IVIG therapy.2
Ig therapy practice is a distinct and complex clinical specialty. Continuous professional advancement through education and certification (IgCN/IgCP) ensures clinical competency and alignment with the national standards. Certification has been shown to improve patient safety and treatment outcomes, reduce errors, minimize staff turnover, and elevate the safety and quality of care delivered to patients receiving Ig therapy.2
Conclusion
Safe and effective IVIG therapy demands early recognition of ADRs, proactive risk mitigation, evidence-based and individualized safe infusion protocols, and timely interventions. Interdisciplinary collaboration is foundational to delivering high-quality care, with nurses, pharmacists, and physicians working together to tailor treatments to individual risk profiles. Incorporating standardized assessment tools and PROMs enables clinicians to objectively monitor IVIG therapy effectiveness, refine care plans, and align treatment with the patient’s evolving goals. This patient-centered approach, supported by shared decision-making and timely therapy adjustments, enhances clinical outcomes and builds trust. The IgNS Standards of Practice provide a legally defensible framework for clinicians, protecting patients, institutions, and healthcare professionals while promoting adherence to best practices. Certification of clinicians in Ig therapy promotes improved clinical outcomes, fewer administration errors, enhanced adherence to safety protocols, and reduced staff turnover, all of which contribute to improved patient safety and organizational performance.
The authors reported no relevant financial disclosures.
References
- Jiang M, et al. Int Arch Allergy Immunol. 2023;184(6):513-528.
- Immunoglobulin National Society. Immunoglobulin Therapy Standards of Practice. Edition 3.2. Accessed September 10, 2025. ig-ns.org/ product/ ig-therapy-standards-of-practice-version-3-2/
- Kato H, et al. Front Immunol. 2021;12:740517.
- Kobayashi RH, Rigas MT. Am J Health Syst Pharm. 2022;79(17):1415-1423.
- Bayry J, et al. J Allergy Clin Immunol Pract. 2023;11(6):1688-1697.
- Allen J, et al. Neurology. 2016;86(16 suppl):P2.269.
- Silvestri NJ, et al. US Neurol. 2015;11(1):40-46.
- Kluzek S, et al. BMJ Evid Based Med. 2022;27(3):153-155.
- Visentin A, et al. Curr Oncol. 2022;30(1):274-283.
- Blood safety. CDC, National Healthcare Safety Network. Accessed September 10, 2025. cdc.gov/nhsn/biovigilance/blood-safety/index.html
- MedWatch: The FDA Safety Information and Adverse Event Reporting Program. FDA. Accessed September 10, 2025. fda.gov/safety/medwatch-fda-safety-information-and-adverse-event-reporting-program
- Martinez C, et al. Clin Exp Immunol. 2021;206(3):384-394.
- Mohamed M. Int J Clin Transfus Med. 2016;4:121-131.
- Shamji MH, et al. Allergy. 2021;76(12):3627-3641.
- Cherin P, et al. Arthritis Rheum. 2002;46(2):467-474.
- Orquera JD, et al. Bull Emerg Trauma. 2022;10(1):44-48.
- Kapoor M, et al. J Neurol Neurosurg Psychiatry. 2022;93(8):876-885.
- Ammann EM, et al. J Thromb Thrombolysis. 2022;53(2):264-272.
- Aggarwal R, et al. Arthritis Res Ther. 2024;26(1):27.
- Kretowska-Grunwald A, et al. J Clin Med. 2022;11(13):3571.
- De Felice ELT, et al. Clin Rev Allergy Immunol. 2024;66(2):241-249.
- Cuesta H, et al. Transfusion. 2022;62(9):1894-1907.
- Esmaeilzadeh H, et al. Clin Immunol. 2021;230:108826.
- Kato H, et al. Front Immunol. 2021;12:740517.
- FDA. Guidance for Industry: Safety, Efficacy, and Pharmacokinetic Studies to Support Marketing of Immune Globulin Intravenous (Human) as Replacement Therapy for Primary Humoral Immunodeficiency. Accessed September 10, 2025. fda.gov/regulatory-information/search-fda-guidance-documents/safety-efficacy-and-pharmacokinetic-studies-support-marketing-immune-globulin-intravenous-human
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Optimizing Intravenous Immunoglobulin Therapy : Enhancing Patient Outcomes Through Iterative and Interdisciplinary Care
Optimizing Intravenous Immunoglobulin Therapy : Enhancing Patient Outcomes Through Iterative and Interdisciplinary Care