Tufts Medical Center
Boston, Massachusetts
The introduction of adalimumab (Humira, AbbVie) biosimilars to the US market in 2023 brought significant changes for providers, patients, and pharmacies. Since its introduction in 2002, Humira has been one of the world’s bestselling drugs for treating inflammatory conditions.1
In November 2023, we published “A Review of Newly Available Humira Biosimilars for Pharmacists” outlining the introduction of the original 9 Humira biosimilars and helpful information for pharmacists. For more background, please refer to the original article.
Pharmacists have been challenged in managing the complexity of switching patients from the reference product to various biosimilars, including navigating clinical appropriateness, education, insurance, affordability, and several practical hurdles. We aim to provide an update on what is new, what we have learned over the past 12 months, and what we are anticipating in the Humira biosimilar market.
Adalimumab Products
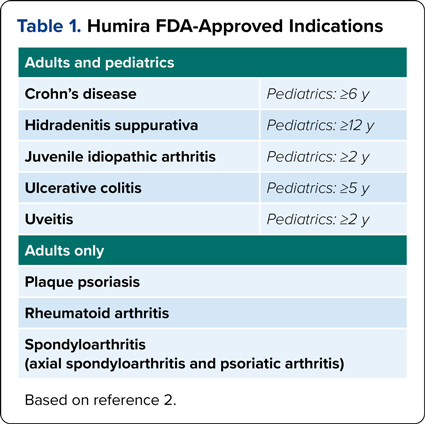
Adalimumab is a tumor necrosis factor-alpha blocking agent that carries many indications for pediatric and adult inflammatory disease states.2 Since our article last year, there has been an additional biosimilar approval in February, adalimumab-ryvk (Simlandi, Alvotech/Teva).3 This product was granted interchangeable status for the citrate-free high-concentration (100-mg/mL) formulation, whereas the initial 2 interchangeable products, adalimumab-adbm (Cyltezo, Boehringer Ingelheim) and adalimumab-afzb (Abrilada, Pfizer), are for the citrate-free low-concentration (50-mg/mL) formulation. Table 1 outlines the indications covered under the reference product Humira.4,5
| Table 1. Humira FDA-Approved Indications | |
| Adults and pediatrics | |
|---|---|
| Crohn’s disease | Pediatrics: =6 y |
| Hidradenitis suppurativa | Pediatrics: =12 y |
| Juvenile idiopathic arthritis | Pediatrics: =2 y |
| Ulcerative colitis | Pediatrics: =5 y |
| Uveitis | Pediatrics: =2 y |
| Adults only | |
| Plaque psoriasis | |
| Rheumatoid arthritis | |
| Spondyloarthritis (axial spondyloarthritis and psoriatic arthritis) | |
| Based on reference 2. | |
Humira and its biosimilars had minor differences in indications last year, including hidradenitis suppurativa (HS) and uveitis. However, now all available biosimilars carry all indications aside from continued pediatric differences for ulcerative colitis (UC), HS, and uveitis (Table 2). The changes included adalimumab-aacf (Idacio, Fresenius Kabi) gaining indication for HS and both Idacio and adalimumab-aaty (Yuflyma, Celltrion) gaining indication for uveitis.6,7 The pediatric differences remain. Pharmacists should continue to carefully evaluate the appropriateness of a product based on the indication for pediatric patients.
| Table 2. Biosimilar Indication Updates Compared With Humira | |
| Biosimilar | Added indication |
|---|---|
| Idacio | Hidradenitis suppurativa |
| Yuflyma | Hidradenitis suppurativa Uveitis |
a Humira only (no biosimilars) for ulcerative colitis in pediatric patients =5 y. b Humira only (no biosimilars) for hidradenitis suppurativa in pediatric patients =12 y. c Humira only (no biosimilars) for uveitis in pediatric patients =2 y. Based on references 6 and 7. | |
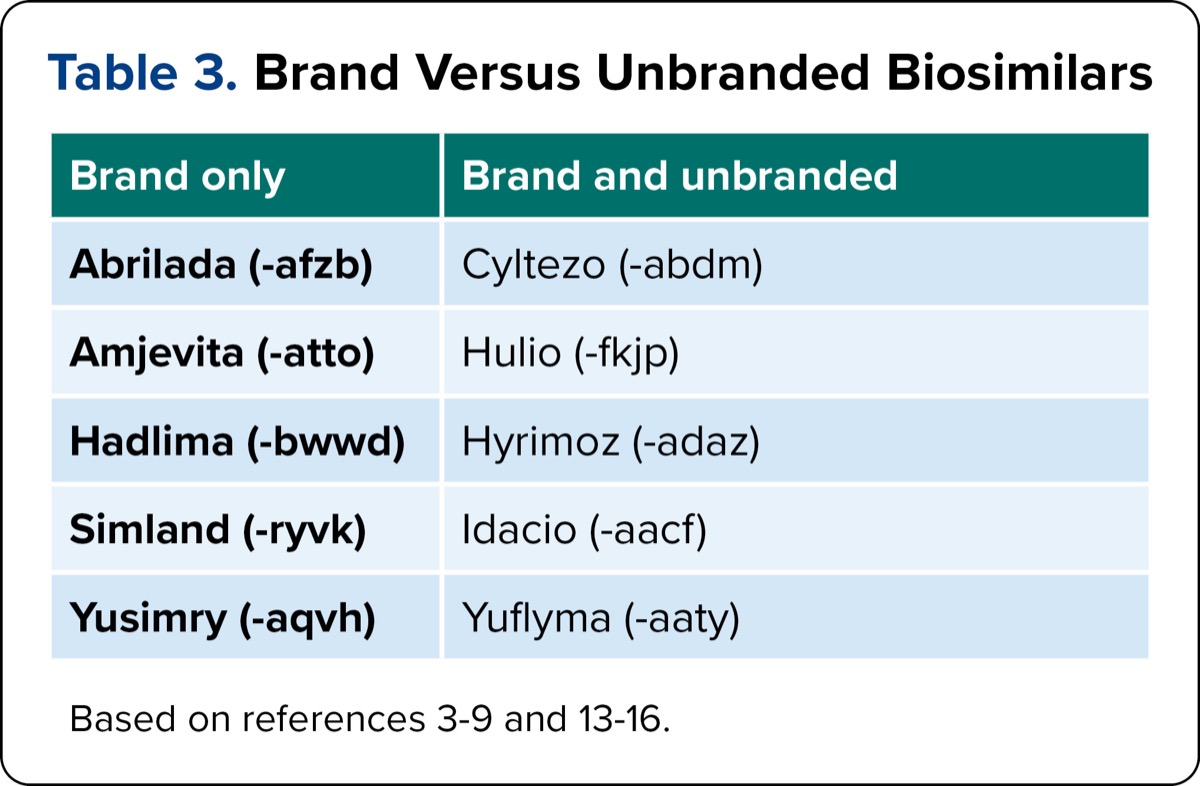
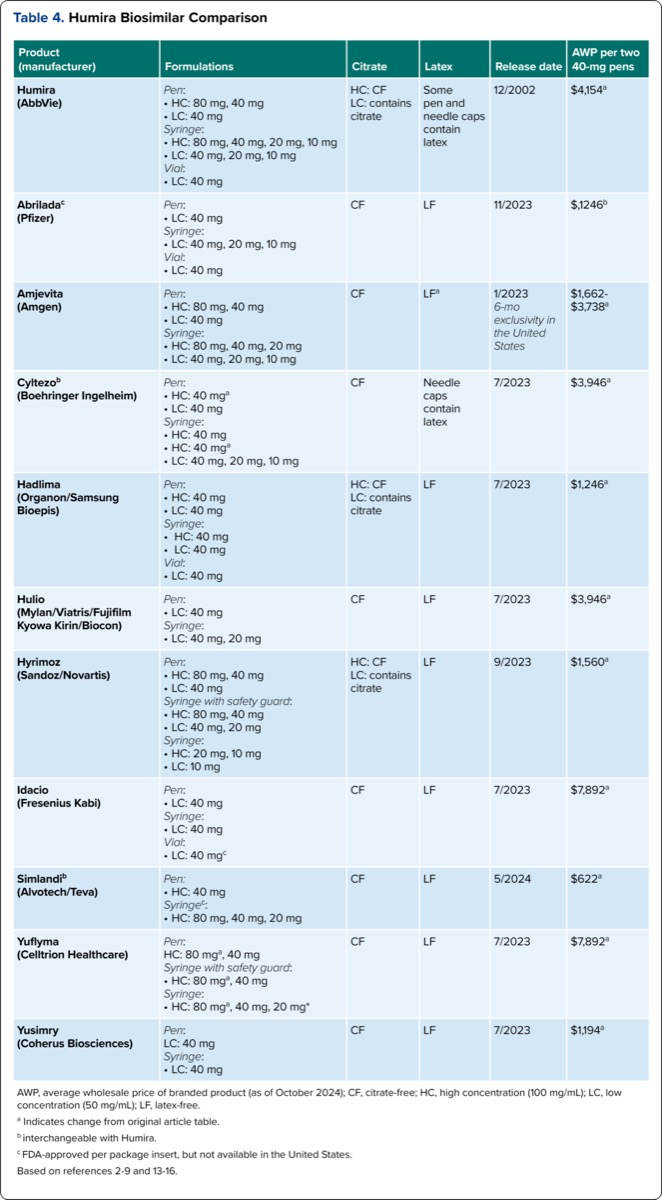
Another factor to consider is the availability of both brand and generic products for certain biosimilars. As outlined in Table 3, 5 of the biosimilars—Cyltezo, adalimumab-fkjp (Hulio, Mylan/Viatris/Fujifilm Kyowa Kirin/Biocon), adalimumab-adaz (Hyrimoz, Sandoz/Novartis), Idacio, and Yuflyma—are available in branded and unbranded forms.4,6-9 The availability of these products has caused challenges for practitioners, patients, and pharmacies addressed in the practical considerations section. Table 4 summarizes key details for the 11 adalimumab products. Changes from the previous table are denoted, including dosage forms, latex content, and average wholesale price.
| Table 3. Brand Versus Unbranded Biosimilars | |
| Brand only | Brand and unbranded |
|---|---|
| Abrilada (-afzb) | Cyltezo (-abdm) |
| Amjevita (-atto) | Hulio (-fkjp) |
| Hadlima (-bwwd) | Hyrimoz (-adaz) |
| Simland (-ryvk) | Idacio (-aacf) |
| Yusimry (-aqvh) | Yuflyma (-aaty) |
| Based on references 3-9 and 13-16. | |
| Table 4. Humira Biosimilar Comparison | |||||
| Product (manufacturer) | Formulations | Citrate | Latex | Release date | AWP per two 40-mg pens |
|---|---|---|---|---|---|
| Humira (AbbVie) | Pen:
| HC: CF LC: contains citrate | Some pen and needle caps contain latex | 12/2002 | $4,154a |
| Abriladac (Pfizer) | Pen:
| CF | LF | 11/2023 | $,1246b |
| Amjevita (Amgen) | Pen:
| CF | LFa | 1/2023 6-mo exclusivity in the United States | $1,662- $3,738a |
| Cyltezob (Boehringer Ingelheim) | Pen:
| CF | Needle caps contain latex | 7/2023 | $3,946a |
| Hadlima (Organon/Samsung Bioepis) | Pen:
| HC: CF LC: contains citrate | LF | 7/2023 | $1,246a |
| Hulio (Mylan/Viatris/Fujifilm Kyowa Kirin/Biocon) | Pen:
| CF | LF | 7/2023 | $3,946a |
| Hyrimoz (Sandoz/Novartis) | Pen:
| HC: CF LC: contains citrate | LF | 9/2023 | $1,560a |
| Idacio (Fresenius Kabi) | Pen:
| CF | LF | 7/2023 | $7,892a |
| Simlandib (Alvotech/Teva) | Pen:
| CF | LF | 5/2024 | $622a |
| Yuflyma (Celltrion Healthcare) | Pen:
| CF | LF | 7/2023 | $7,892a |
| Yusimry (Coherus Biosciences) | Pen:
| CF | LF | 7/2023 | $1,194a |
| AWP, average wholesale price of branded product (as of October 2024); CF, citrate-free; HC, high concentration (100 mg/mL); LC, low concentration (50 mg/mL); LF, latex-free. a Indicates change from original article table. b interchangeable with Humira. c FDA-approved per package insert, but not available in the United States. Based on references 2-9 and 13-16. | |||||
Clinical and Safety Considerations
Since the implementation of the adalimumab biosimilar products into clinical practice in the United States, there has been limited real-world evidence evaluating clinical and/or safety implications on patients’ disease control. Data often must be extrapolated from other global areas with more experience utilizing adalimumab biosimilars, such as Europe and Canada.
An observational study conducted in Canada sought to evaluate real-world experience on the comparative safety and efficacy of adalimumab biosimilar therapy after a mandatory nonmedical switch.10 The analysis included 191 patients with inflammatory bowel disease (IBD) receiving Humira, 145 of whom underwent the mandated switch to a biosimilar agent, Idacio. Adverse events including rash, nausea, or vomiting were reported in 10 patients in the biosimilar group, and 7 patients required a switch back to the reference product, Humira. From an efficacy standpoint, there were no differences in inflammatory biomarkers (C-reactive protein and fecal calprotectin) measured before and after the biosimilar switch, and when compared with the patients who stayed on Humira (P=0.48 and P=0.142, respectively). No IBD-related emergency department visits or hospital admissions occurred. The authors concluded that the clinical benefit of Humira was sustained after a nonmedical switch to an adalimumab biosimilar.10
Similarly, Jin et al reported real-world experience of adalimumab-atto (Amjevita, Amgen) biosimilar use in patients with IBD in 6 countries in Europe.11 This observational study included 2 cohorts: 239 patients who initiated Amjevita as first-line advanced therapy and 136 patients who were switched to Amjevita from the reference product (Humira). Patients were on treatment for a median of 7.5 months at the time of analysis. Approximately 75% of patients who initiated Amjevita as first-line advanced therapy and 89% of those who switched from the reference product were reported to be in clinical remission by their physicians. Patients reported high rates of satisfaction with the biosimilar product, with minimal impairment of healthcare-related quality of life, utilizing various self-assessment tools. This finding suggests that the patient experience associated with the use of Amjevita appears similar to that of the reference product, regardless of prior exposure.11
Data denoting the successful use of adalimumab biosimilars in other global areas have been helpful in reassuring patients and providers that they should expect minimal, if any, clinical or safety differences when switching from the reference product. However, there are downsides to extrapolating these data to US populations. Many of the published reports are in singular disease states and cannot always be generalized to all indications. Differences in insurance plans and formularies across countries also may confound these findings.
Despite the positive findings shown in international real-world data, adalimumab biosimilars in the United States remain underutilized, likely due to varying patient and provider comfortability with Humira, and apprehensions toward switching to a biosimilar. One recent study found that approximately 98% of Medicare Part D prescription plans covered Humira, while only 53.4% of plans covered at least 1 biosimilar, including about 60 plans that paid only for biosimilars, excluding Humira. Most of the biosimilars covered had a wholesale acquisition cost close to that of Humira’s nearly $7,000 monthly list price. The researchers noted that narrow formulary coverage could limit adoption of Humira biosimilars.12
Prescribing rates also remain heavily driven by insurance formulary restrictions. Pharmacists can play a key role in providing education about biosimilars to address these concerns and contribute to healthcare savings.
Practical Considerations
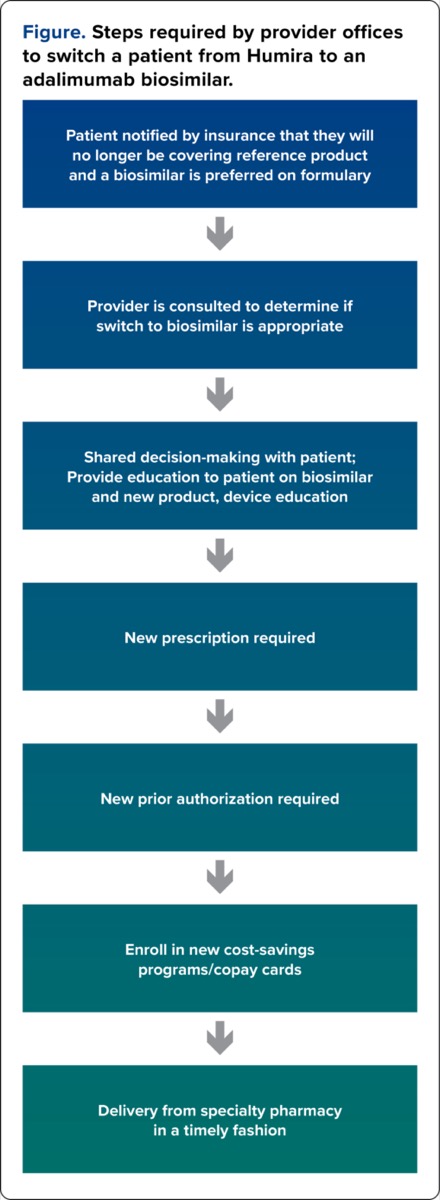
The Figure demonstrates the steps generally required by the provider office to switch a patient from Humira to an adalimumab biosimilar. Pharmacists are likely to be heavily involved in this workflow, so it is imperative that they understand and become proficient in this process.
| Patient notified by insurance that they will no longer be covering reference product and a biosimilar is preferred on formulary | ||
| Provider is consulted to determine if switch to biosimilar is appropriate | ||
| Shared decision-making with patient; Provide education to patient on biosimilar and new product, device education | ||
| New prescription required | ||
| New prior authorization required | ||
| Enroll in new cost-savings programs/copay cards | ||
| Delivery from specialty pharmacy in a timely fashion | ||
| Figure. Steps required by provider offices to switch a patient from Humira to an adalimumab biosimilar. | ||
Patients are often notified by their insurance company when their formulary is changing, and a biosimilar will become the preferred product. In some instances, however, patients may be unaware of this change, and they are notified by the filling pharmacy that Humira is no longer covered by their insurance. Patients will most often contact their provider’s office for next steps. The ordering provider should be consulted to determine whether switching to an adalimumab biosimilar is appropriate, taking into consideration the patient’s disease severity or other patient-related factors. Shared decision-making should take place between the provider, pharmacist, and patient to ensure patients are comfortable and well informed of biosimilar therapy.
Switching to an adalimumab biosimilar has proven more complicated than just sending a new prescription to the pharmacy. Once patients agree to switch, several steps still need to take place to ensure patient understanding, medication affordability, and timely dispensing of the medication. Pharmacists are in an excellent position to provide education on adalimumab biosimilars to the patient. This includes training on new self-injection devices, since all the adalimumab biosimilars have different products with various doses and methods of delivery. From a coverage perspective, there has not been consistency across insurance plans whether a new prior authorization (PA) will be required for the adalimumab biosimilar. Some insurance plans transfer the active Humira PA to cover the biosimilar, while others require a new one. This may be a source of delay in initiating the biosimilar. Lastly, patients who utilize cost assistance programs or copay cards will need to be enrolled in the respective manufacturer-specific programs. All the above processes should be completed to ensure timely delivery of the adalimumab biosimilar from the specialty pharmacy, and to allow for continuity of care with minimal interruptions in treatment.
In addition to the complex workflow in switching patients to a biosimilar, several other practical challenges persist. The different product sizes available may continue to affect new-start adalimumab patients requiring loading doses, and therefore more injections, compared with the reference product with larger strengths available. As additional biosimilars are approved, electronic health records will need to continually be assessed and updated to include the entries required for prescriptions or in-clinic administrations, along with staff and nursing education. The introduction of brand and generic products for certain biosimilars has also added to confusion among electronic prescriptions and PAs. In some instances, prescriptions and PAs may be inconsistent, causing inability to dispense to patients and lead to delays in care. For example, if a PA is in place for the brand name biosimilar, but a generic prescription is sent, the pharmacy may receive a rejected claim. Lastly, shortages and back orders of products have caused potential delays in care, especially with changes to insurance formularies leading to large patient populations requiring the same product simultaneously.
The overall goal of biosimilars is to increase competition, lower costs, and ultimately lessen the financial burden on patients and healthcare systems. Throughout 2024, we have seen insurance formularies adjust to prefer biosimilars; however, the financial impact is still unknown and highly anticipated data throughout the healthcare industry.
Conclusion
The introduction of adalimumab biosimilars over the past 2 years has presented an opportunity for cost savings, as well as a unique challenge for pharmacists, healthcare teams, and patients. In the past 12 months, the adalimumab biosimilar market has introduced a 10th biosimilar, Simlandi, as well as some indication updates. From a clinical outcome and safety perspective, all the adalimumab products continue to exhibit similar effects. However, key practical differences, such as self-injection device type, citrate content, and insurance coverage, can help guide pharmacists and patients to choose the best product. Promoting utilization of biosimilars is an important initiative for pharmacists to maximize healthcare savings for patients and healthcare systems. Moving forward in the biosimilars landscape, it will be crucial to evaluate and assess the real-world cost savings observed for the healthcare system and patients. Insurance companies continue to dictate which preferred product patients will use, but the tools mentioned above can help providers and patients understand the change to maximize success. Pharmacists are well equipped to help navigate this change from a clinical, practical, and financial perspective and ensure that patients continue or begin safe and effective therapy for their complex conditions.
The authors reported no relevant financial disclosures.
References
- Coghlan J, He H, Schwendenman AS. Overview of Humira biosimilars: current European landscape and future implications. J Pharm Sci. 2021;110(4):1572-1582.
- Humira (adalimumab) [package insert]. AbbVie; 2024.
- Simlandi (adalimumab-ryvk) [package insert]. Alvotech/Teva; 2024.
- Cyltezo (adalimumab-adbm) [package insert]. Boehringer Ingelheim; 2024.
- Abrilada (adalimumab-afzb) [package insert]. Pfizer; 2023.
- Idacio (adalimumab-aacf) [package insert]. Fresenius Kabi; 2024.
- Yuflyma (adalimumab-aaty) [package insert]. Celltrion; 2023.
- Hulio (adalimumab-fkjp) [package insert]. Mylan/Viatris/Fujifilm Kyowa Kirin/Biocon; 2023.
- Hyrimoz (adalimumab-adaz) [package insert]. Sandoz/Novartis; 2024.
- Bedi H, Rosenfeld D, Hoang T, et al. A175 real-world experience on switching from adalimumab to biosimilars in patients with inflammatory bowel disease—an observational study from three tertiary care centers. J Can Assoc Gastroenterol. 2023;6(1):19-20.
- Jin R, Nduka C, Courmier D, et al. Real-world experience of adalimumab biosimilar (ABP 501) use in patients with inflammatory bowel disease in Europe. Adv Ther. 2024;41(1):331-348.
- Klebanoff MJ, Li P, Lin JK, et al. Formulary coverage of brand-name adalimumab and biosimilars across Medicare Part D plans. JAMA. 2024;332(1):74-76.
- Amjevita (adalimumab-atto) [package insert]. Amgen; 2023.
- Hadlima (adalimumab-bwwd) [package insert]. Organon/Samsung Bioepis; 2024.
- Yusimry (adalimumab-aqvh) [package insert]. Coherus Biosciences; 2023.
- Adalimumab. In: Lexi-Drugs. Lexicomp Online; 2021. Updated October 18, 2024. Accessed November 6, 2024. https://online.lexi.com
Copyright © 2024 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Pharmacists’ Key Role in The Management of Humira Biosimilars
Pharmacists’ Key Role in The Management of Humira Biosimilars