A growing alternative to traditional compounding is making its debut in Europe and the United States.
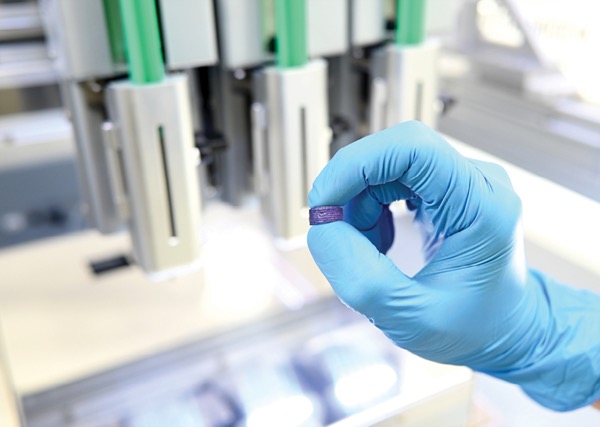
The London-based biotech FABRX sells pharmaceutical 3D printers that produce compounded medications in clinics or hospitals, based on a pharmacist or physician prescription. FABRX printers can also prepare personalized medicines for use in clinical trials (see sidebar). The devices represent one form of distributed manufacturing, a broad term that encompasses technologies such as 3D printing or the protein development process used by Massachusetts-based Sunflower Therapeutics.
“The starting point for personalization is the dose,” said Anna Worsley, PhD, the CEO of FABRX AI, a Spanish subsidiary of FABRX. The size of the 3D-printed tablet is directly proportional to the dose, she explained. “You calculate the concentration and formation before you put it into the printer; our software can do it automatically.” The printers include scales to ensure that tablets are the proper weight, and a spectrometer for chemical analysis.
Dr. Worsley, who spoke about FABRX in a session at the ASHP Midyear 2024 Clinical Meeting & Exhibition, in New Orleans, noted that the company offers three days of training for every organization that purchases a printer, along with ongoing technical support.
“People can often learn how to use the printers in an afternoon. But we offer the extended training to provide additional assurance,” Dr. Worsley said. FABRX offers two printers: one with a single printhead and a larger model with three printhead attachments. The three-printhead model is better suited for multidrug medicines, or polypills, she explained.
Customization options include the ability to produce easy-to-swallow capsules, chewables and flavorings that improve the taste of the drug. This can be especially helpful for children who refuse to take medications with an unpleasant taste, Dr. Worsley noted.
Compounding pharmacies have long developed personalized medicines, Dr. Worsley pointed out—that’s the entire point of compounding, to create a bespoke medication because a standard product will not work for a given patient. But the traditional compounding process is labor-intensive.
“Compounding involves processes like crushing tablets and mixing powders together,” Dr. Worsley said. This manual approach can cause inadvertent errors or potentially expose a technician to hazardous residues. Compared with that, Dr. Worsley added, compounding via 3D printing can be safer and faster; in a study recently published by FABRX, their printers reduced manual labor by 55% versus traditional methods, with 35% cost savings reported for some medications (Int J Pharm 2025;671:125251).
FABRX printers are mostly used in Europe; one client is Gustave Roussy Institute, an oncology hospital based in Paris with two printers on hand. The company is starting to expand into the United States with printers already being installed.
Dr. Worsley noted that many new players entered the market in 2023, the year FABRX launched its multi-head printer. Although competition presents challenges in gaining market share, it is a good sign in Dr. Worsley’s view. “In a way it benefits us because it shows overall growth in the field.”
Regulatory Innovation Needed
J. Christopher Love, PhD, a professor of chemical engineering at Massachusetts Institute of Technology, in Cambridge, and co-founder of Sunflower Therapeutics, noted that “there are several examples of technologies designed for distributed manufacturing of medicines. The proof of concept has been established,” he said, adding that the biotech company offers manufacturing systems to produce proteins on demand for products such as vaccines.
“The idea is that anyone anywhere should be able to produce proteins for their applications,” Dr. Love said, reducing the overhead and lead time of traditional drug manufacturing.
Ten years ago, he noted, technologies for distributed manufacturing were only in the lab; indeed, lab work was the roots of Sunflower Therapeutics, which obtained early funding from the Department of Defense. Other companies in this space include Cellares, On Demand Pharmaceuticals, Quantoom and Univercells.
Currently, proteins, cell therapies, viral vaccines and mRNA can all be made with “small footprint manufacturing solutions,” Dr. Love said. He continues to refine the biology used for production to make proteins, such as monoclonal antibodies used for cancer treatment.
While the science is largely settled, hurdles to widespread distributed production remain.
“The question today is, how do we transform a manufacturing base from one that is highly centralized and large-scale to something that can rely on a robust distributed network?” Dr. Love said. “This is a key question for both regulatory science and public–private partnerships to address.”
Pharmacy Technology Report requested an interview with FDA officials about the agency’s stance regarding the potential benefits and any safety risks of 3D-printed medications. The FDA declined an interview but referred us to a general landing page about the agency’s view of 3D-printed materials. That page links to technical guidance documents dating back to 2016, and notes that there are specific considerations for printing a medical device, biologic or drug. The agency recommends manufacturers consult guidance documents relevant to their product.
The sources reported no relevant financial disclosures beyond their stated employment.
This article is from the September 2025 print issue.