Much of the conversation about temperature management and cold chain in pharmacy distribution has focused on the “last mile,” that is, the delivery of products such as medications and vaccines from the transportation hub to the end user. But for a hospital or other medical facility responsible for dispensing and/or administering those products, there’s another challenge: the “last foot.”
Temperature-sensitive medications travel a winding path, moving all the way from manufacturers to distributors, hospitals and ultimately to pharmacies, noted Amardeep Chahal, the senior vice president for global marketing, strategy and corporate development with Cold Chain Technologies. In many cases, those medications are then shipped from pharmacies to patients. And that’s where it gets tricky.
“While there has been a lot of attention on compliance with temperature control in the first and last mile of this process, driven by regulatory authorities, there has been less oversight on the last foot between the medication and the patient,” Mr. Chalal said. “As more and more medications need temperature control, pharmacies need help in defining the procedures they need to follow and structures they must have in place.”
To further define what those procedures should be, in 2022 and 2023, ASHP worked with Cold Chain Technologies to sponsor a series of three executive forums on cold chain management of pharmaceuticals. More than 50 people from health systems and other key stakeholder organizations attended.
Forum participant Jason Chou, PharmD, the vice president of pharmacy services for Louisiana-based Ochsner Health System, underlined a key takeaway in an interview with Pharmacy Technology Report. “There’s a misconception that everybody in the cold chain continuum understands what everybody upstream is doing,” he said. “The deliveries we receive can be very complicated. There’s still a lot to be desired in terms of accessibility to that data for those of us who are downstream.”
There isn’t a better recent example of complicated cold chain therapeutics than the need for rapid distribution of ultra–cold chain vaccine products during the height of the COVID-19 pandemic. The vaccines dramatically accelerated understanding of the importance of processes and systems needed to ship and store these products, an aspect of cold chain management that is likely to expand with the increasing number of approved cell- and gene-based therapies that also require such specialized handling, Mr. Chahal noted.
“COVID-19 put a spotlight on the need for solid cold chain infrastructure within pharmacies that had previously been missing,” he said. “More and more drugs that are getting launched require –20° C and lower conditions.”
Dr. Chou agreed that ultra–cold chain logistics will continue to challenge health-system pharmacies, partly because the most sensitive and costly cold chain medications sometimes come in through channels that may experience longer delays in final disposition than other drugs. “Our traditional wholesale orders are typically delivered straight to our pharmacy and are difficult to miss,” he said. “But the really expensive, vulnerable medications come from hubs or limited distribution sources that aren’t [shipped] with the vast majority of our typical orders. Those are the ones that are of the most concern for tracking and timeliness of getting that product in and stored appropriately.”
As an example, “if a truck delivers a pallet of medications to our loading dock, we may not find it for a certain period of time,” Dr. Chou said. “So, it’s important that we know not only if the package is good from the point it arrives at the dock, but what if it sits there for two or three hours?”
In many of these last-mile spaces, low-cost solutions often are used because of tight margins, Mr. Chahal said. That includes using a calendar and either adding or reducing refrigerants to seasonal solutions for temperature protection when shipping to warmer or colder destinations. “But improved intelligence tools, like CCT Route Pro [bit.ly/49fQIqe], are available to help determine the right pack-out configuration given a package’s origin, destination and the route it’s going to take, so there is less guessing involved.”
Dr. Chou said the ideal system would involve some level of delivery tracking and logging for all temperature-sensitive medication shipments, including those that must remain at room temperature, up to the point of delivery with some level of delivery confirmation. “To me, we shouldn’t stop tracking until the medication is unpacked and put in its final destination, appropriately stored.”
Adding sophisticated, WI-Fi–enabled data loggers that can track precise physical location and provide real-time notification of excursions to most drug shipments would greatly increase cost at comparatively little benefit. “It has probably been in some capacity cheaper just to waste products that ended up having excursions,” Dr. Chou said.
Higher-Cost Solutions
Many of the novel drugs that require ultra-cold handling also have higher price tags and rapid-access needs that warrant higher-cost cold chain solutions. For example, the spinal muscular atrophy gene therapy onasemnogene abeparvovec-xioi (Zolgensma, Novartis), which is shipped at –60° C [–76° F] or lower, costs approximately $2.1 million and has been found to be most effective when given to presymptomatic infants whose disease duration was eight months or less before receiving therapy (Pediatr Neurol 2023:144:60-68). “The stakes are a lot higher with drugs like these,” Dr. Chou said. “For these drugs, we need what we had with the COVID vaccines: real-time shipment information, as well as location and temperature tracking up until the point we put them into ultra-cold storage.”
Many health systems will need to address knowledge gaps regarding cold chain handling and logistics, Mr. Chahal noted. “I have visited many pharmacies where technicians or nurses are required to bring in the boxes, check on their temperature control status, pack them out and ensure that they are stored and conditioned appropriately,” he said. “That’s not something they have ever been trained to do and so they often are learning on the fly. They want to do the best, but they need support.”
That was the primary impetus behind the ASHP forums, he said. One important resource that emerged from those forums was a self-assessment tool, designed to help health systems understand their own level of readiness around several key cold chain operational areas, including:
- education and training;
- emergency preparedness;
- monitoring;
- policies and procedures;
- receipt and storage; and
- shipping and transport.
For example, does your organization have protocols that define allowable temperature excursions? Are all drugs received in the pharmacy and controlled from the point of receipt? Is there a limit to higher-cost drugs stored in smaller sites and clinics? (The tool can be accessed in the second forum report at bit.ly/4ctCNjc.)
“Various experts working together with ASHP have developed this checklist and [it] is a great resource for all pharmacies around the country to strengthen their cold chain infrastructure and processes,” Mr. Chahal said. “This should be used in training, and possibly offered as part of accreditation or CE [continuing education] hours. With a growing number of medications requiring cold chain and the impact that proper transportation and storage can have on the medication and eventually on patients’ lives, we can’t lose momentum.”
The sources reported no relevant financial disclosures beyond their stated employment.
More Supplier Perspectives
It’s an unprecedented time for temperature-controlled shipping. While hospitals and health systems are increasingly providing highly sensitive, ultra–cold chain products such as cellular and gene therapies (CGTs), hundreds of thousands of doses of glucagon-like peptide-1 (GLP-1) agonists and erectile dysfunction medications are also being shipped not only to pharmacies but directly to patients’ homes. All these medications require specialized and individualized solutions not only in temperature-controlled packaging, but in the integrated, highly visible systems that track location and excursions.
Packaging Solutions
“We have a worldwide patent on one-piece construction for liners—a liner that substitutes for multiple components to the package, alleviates thermal gaps, and is easier to inventory and pack out,” said Jeffrey Griffin, the director of business development and sales at Coldkeepers. Their U.S.-made Curbside Recyclable Paper One-Piece Thermal Insulated Box Liner ships flat and is utilized for ambient, fresh, refrigerated, and frozen products between 24 and 72 hours, with gel packs or dry ice to extend transit times.
The biggest growth area Coldkeepers has seen within the past year is in its thermal insulated mailers, designed to fit inside FedEx and UPS Express boxes. “503A compounders are able to use these along with a couple of gel packs to ship vials of the GLP-1 [agonist] medications,” Mr. Griffin said. “One pharmacy that we work with had five employees and one pharmacist two years ago. Now they have 600 employees and 24 full-time pharmacists, and they buy three or four truckloads of product per week from us.”
Ontario-based TempAid also has a thermal insulated shipping mailer, SpeedyPac, with a proprietary phase-change material integrated into the lining to eliminate the need for EPS coolers, a corrugated box and separate refrigerants. “These are the original and still the only phase-change injected mailers. They come preloaded, grab and go, so you don’t have to pack any frozen parts,” said Joe Futino Jr., the vice president of sales. “Because every manufacturer has a different temperature range for these shipments—Ozempic [semaglutide injection, Novo Nordisk] is cold chain while other medications move at a mix of ambient and cold temperatures—we had to do a lot of work in developing these specialty phase change materials.”
For ultra–cold chain products, TempAid has designed solutions around dry ice shipping loaded into vacuum insulated panel boxes (VIPs) called Steadipaks. “Dry ice cannot be shipped by air, and our Steadipak boxes allow you to ship ground nationwide with less dry ice and two to four times the performance, and still reuse the box afterward,” Mr. Futino said. “That’s because of our patented inner tote liner which weighs less than a pound and limits air loss. You can slide these liners out of the box, take them into the freezer with the dry ice, reload them and send them back out.”
The inner liners also facilitate heavy reuse of the boxes themselves. “A vacuum panel’s pain point is when it becomes damaged and unusable due to microabrasions from the dry ice,” Mr. Futino said. “The inner liner protects the boxes, giving them an extra-long duration and life cycle. We can get 144 hours of performance on a single VIP.”
Tracking Solutions
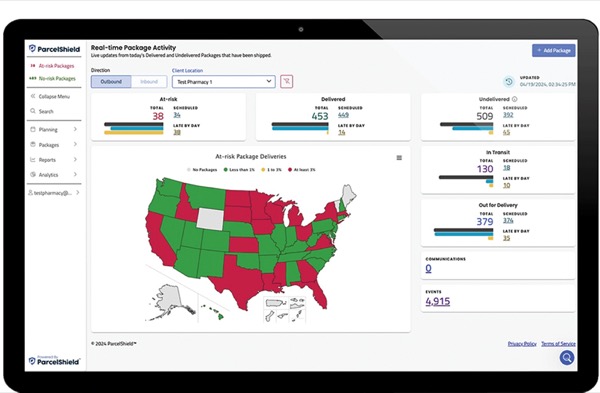
Many ultra–cold chain products are also costly, such as atidarsagene autotemcel (Lenmeldy, Orchard Therapeutics), launched in March 2024 for the treatment of metachromatic leukodystrophy. It’s shipped in the vapor phase of liquid nitrogen at less than –130°C (–202°F) and has a wholesale acquisition cost of $4.25 million (qrs.ly/r6g3u4d). That makes real-time tracking of these shipments, their temperature and other conditions essential. “They are not only time-sensitive and temperature-sensitive, but they can be sensitive to shock, how they are handled, even if they are kept in upright conformations,” said William Gatlin, PharmD, director of customer success at ParcelShield, whose advanced visibility and tracking platform monitors more than 62 million prescription deliveries annually, driven by predictive analytics and machine learning. “We can track all of this across the entire time line from the manufacturer, through the pharmacy, to the patient for therapy, and then continue to have that information for years to come as clinicians continue to monitor the patients.”
With the growth of CGTs that require patient-specific cells to be shipped to the pharmaceutical company for drug manufacturing, two-way logistics has become increasingly important. “This is new for many hospitals and health systems,” said Neil Garnichaud, the chief product and technology officer for ParcelShield. “Following apheresis, they must send the specimens out to the pharmaceutical company in a highly controlled fashion, before the medication is returned to them. So, they need to engage carriers that are properly equipped to do that and monitor them on the outbound side as well. How do you make sure those carriers are in the right place? How do you ensure with double coverage that there’s a courier available to pick up a shipment?”
Using ParcelShield’s integrated dashboard, hospital pharmacies can:
- identify optimal carriers and modes for on-time delivery;
- use machine learning to avoid disruptions and minimize costs;
- get recommendations for reliable carriers and rates; and
- pinpoint where shipments might encounter risks.
“Our solution also allows you to ultimately track the outcomes, because so many of these therapies have complicated payment structures,” Mr. Garnichaud said. “If a therapy fails, you’ll need to conduct a retrospective analysis on the logistics to determine if the handling was appropriate and timely—from the outgoing sample process to the entire treatment journey.”
—Gina Shaw
The sources reported no relevant financial disclosures.
This article is from the September 2024 print issue.