In 2024 on-site surveys, The Joint Commission will look to make sure hospital and critical access pharmacies have policies and procedures to meet the 2022 updates to USP General Chapter <797> for sterile compounding, which became effective Nov. 1, 2023.
Robert Campbell, PharmD, BCSCP, the senior director of the Standards Interpretation, Accreditation Decision Management and Medication Safety Division of Accreditation and Certification Operations for The Joint Commission, provided several tips to meet these requirements during a session at the ASHP 2023 Midyear Clinical Meeting & Exhibition, in Anaheim, Calif.
He cautioned that his comments offered guidance on Joint Commission evaluation processes for sterile nonhazardous and hazardous medication compounding. State laws, in contrast, may have more restrictive requirements, so that following only updated Joint Commission guidelines may put hospital and critical access programs out of state compliance.
Healthcare organizations should develop and implement policies and procedures for the most stringent requirements, he stressed.
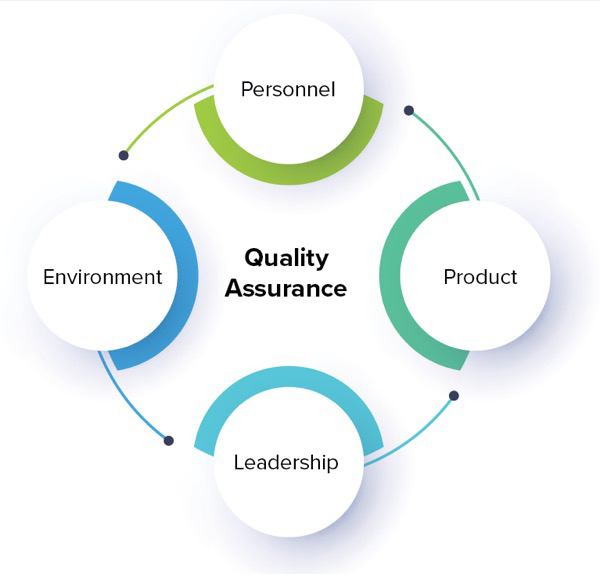
The Joint Commission has always had four quadrants of components for surveyors to review: personnel, product, environment and leadership, Dr. Campbell said. Under the new provisions, medications drawn up in a syringe and administered immediately to a patient, coupling a medical vial to a bag with a marketed coupling device and immediately administering that product, and preparing alteplase for use in the emergency department is not considered compounding by The Joint Commission, Dr. Campbell said. However, adding a medication to an IV bag and administering that to a patient, and drawing up medication in a syringe in anticipation of needing it during a surgical procedure, is considered compounding.
In revisions to Section MM.05.01.07, the first element of performance was reworded but the intention is the same, he said. The language stated that a pharmacist should supervise all compounding of drugs and biologicals except in urgent situations. The supervising person does not have to be a pharmacist, Dr. Campbell noted, but the pharmacy is the location that should compound all drugs known to be needed by the surgical suite or other areas of the hospital during nonemergencies.
For elements 2-6, Joint Commission surveyors will rely on pharmacy personnel following their hospital’s policies and procedures. As an example, element 2 requires hospitals to develop policies for sterile medication compounding in accordance with state and federal laws. If a surveyor finds air pressures are off, and a hospital doesn’t have a policy for that, it will be cited.
Regarding element 4, maintaining a proper environment, “The Joint Commission is only going to survey to the ‘musts’ and ‘shalls’ of USP <797>,” Dr. Campbell explained. “If it has a ‘must’ or a ‘shall,’ that is what we’re going to hold you accountable to. All the ‘coulds,’ ‘shoulds,’ ‘mights,’ ‘mays,’ ‘considers,’ that is for the healthcare organization to decide.”
However, hospitals should carefully implement policies and procedures to ensure they can be followed, he said. Some 65% of The Joint Commission’s findings have been based on individual hospital policies and procedures, not on the group’s standards.
Notable changes to the 2022 version of Chapter <797> include:
- Risk levels are now referred to as categories. Category 1 compounded sterile products (CSPs) include those prepared in a primary engineering control located in a segregated compounding area. Beyond-use dates (BUDs) are 12 or fewer hours at controlled room temperature or 24 or fewer hours when refrigerated. Category 2 CSPs must be prepared in a cleanroom suite and may be assigned a BUD as defined in the chapter.
- Compounders and designated people must complete all competencies in maintaining quality, principles and practices, garbing, and media fill. This group includes personnel providing final verification located remotely. Hospitals can decide what competencies need to be maintained for other workers entering the rooms; however, training and competency assessment must occur at least once every three years. Some states require annual training for nurses. Competencies surveyors will look for include proper hand hygiene, garbing, cleaning and disinfecting, and aseptic technique.
- Visual observation of hand hygiene and garbing, gloved fingertip and thumb sampling, and media-fill testing have been increased. These observations now should be completed every six months instead of annually for Category 1 and 2 CSPs and every three months instead of semiannually for Category 3 CSPs. The Joint Commission is providing hospitals six months to transition to this new competency schedule.
- Garbing now should be completed using low-lint products. If using a restricted access barrier system (RABS), disposable gloves should be worn inside the gloves attached to RABS sleeves.
- Viable air sampling needs to be conducted every six months for Category 1 and 2 CSPs and monthly for Category 3 CSPs. Surface sampling needs to be conducted monthly for Category 1 and 2 CSPs and weekly for Category 3 CSPs. People conducting in-house viable sampling must have training and competencies in how to collect, read, store and incubate samples. Incubators and warmers must be monitored and properly maintained.
- Healthcare organizations must ensure competencies that are completed for immediate-use compounding. These competencies have to be completed regardless of the BUD used.
- There must be a written quality assurance program. The program should be created to identify adherence to policies and procedures, prevention and detection of errors, and quality issues and more.
The Joint Commission will maintain resources on its website for the 2008 and 2022 versions of Chapter <797> through Jan. 1, 2025, Dr. Campbell said. The website (www.jointcommission.org/standards/standard-faqs) has an FAQs section where people can sign up for alerts about new posts.
Dr. Campbell reported no relevant financial disclosures.
This article is from the June 2024 print issue.