Originally published by our sister publication Infectious Disease Special Edition
Bionpharma issued a voluntary nationwide recall of atovaquone oral suspension, 750 mg/mL because of bacterial contamination.
Atovaquone is a quinone antimicrobial drug indicated to prevent Pneumocystis jirovecii pneumonia in adults and adolescents 13 years of age and older.

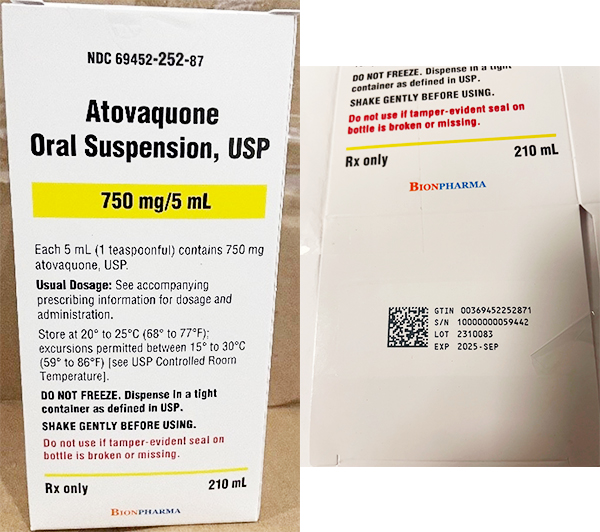
A single batch (2310083) of atovaquone oral suspension, 750 mg/mL was being recalled. The affected batch of the product was manufactured at CoreRx Inc. and shipped nationwide to customers between Dec. 21, 2023, and June 20, 2024, and distributed through wholesalers and retailers.
The product was found to be contaminated with Cohnella bacteria.
“In the population most at risk, immunocompromised population, there is a reasonable probability that microbial contamination of Atovaquone Oral Suspension can result in disseminated, life-threatening infections such as inflammation of the heart and permanent damage to soft tissue,” the company said in a statement. However, Bionpharma has not received any reports of adverse events related to this recall so far.
The company is notifying its distributors and customers by email and arranging for return/replacement etc. of the recalled batch of the product. Distributors/retailers that have affected lot of the drug product that is being recalled should immediately cease distribution and remove it from their active inventory. Consumers who have the affected lot of the product should stop using it and return to the place of purchase.
Consumers with questions regarding this recall can contact Bionpharma by phone at (888) 235-2466 (Mon-Fri 9 a.m.-5 p.m. EST) or via email to drugsafety@bionpharma.com. Consumers should contact their physicians or healthcare providers if they have experienced any problems that may be related to taking or using the affected lot of the drug product.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail or by fax (800-332-1088).
The recall includes the following product and batch number:
| Production date | Release date | Product name | NDC | Lot No. | Expiration date |
| Oct. 26, 2023 | Dec. 5, 2023 | Atovaquone oral suspension | 69452-252-87 | 2310083 | September 2025 |

