Originally published by our sister publication Specialty Pharmacy Continuum
- First ready-to-use subcutaneous formulation of bortezomib now available
- Marks Amneal’s fourth 505(b)(2) injectable product launch in the last year
Amneal Pharmaceuticals Inc., a global biopharmaceutical company, and Shilpa Medicare Limited announced the U.S. launch of Boruzu, a new presentation of bortezomib for ready-to-use subcutaneous or IV administration. This new ready-to-use oncology product reduces the compounding preparation steps typically required with administration.
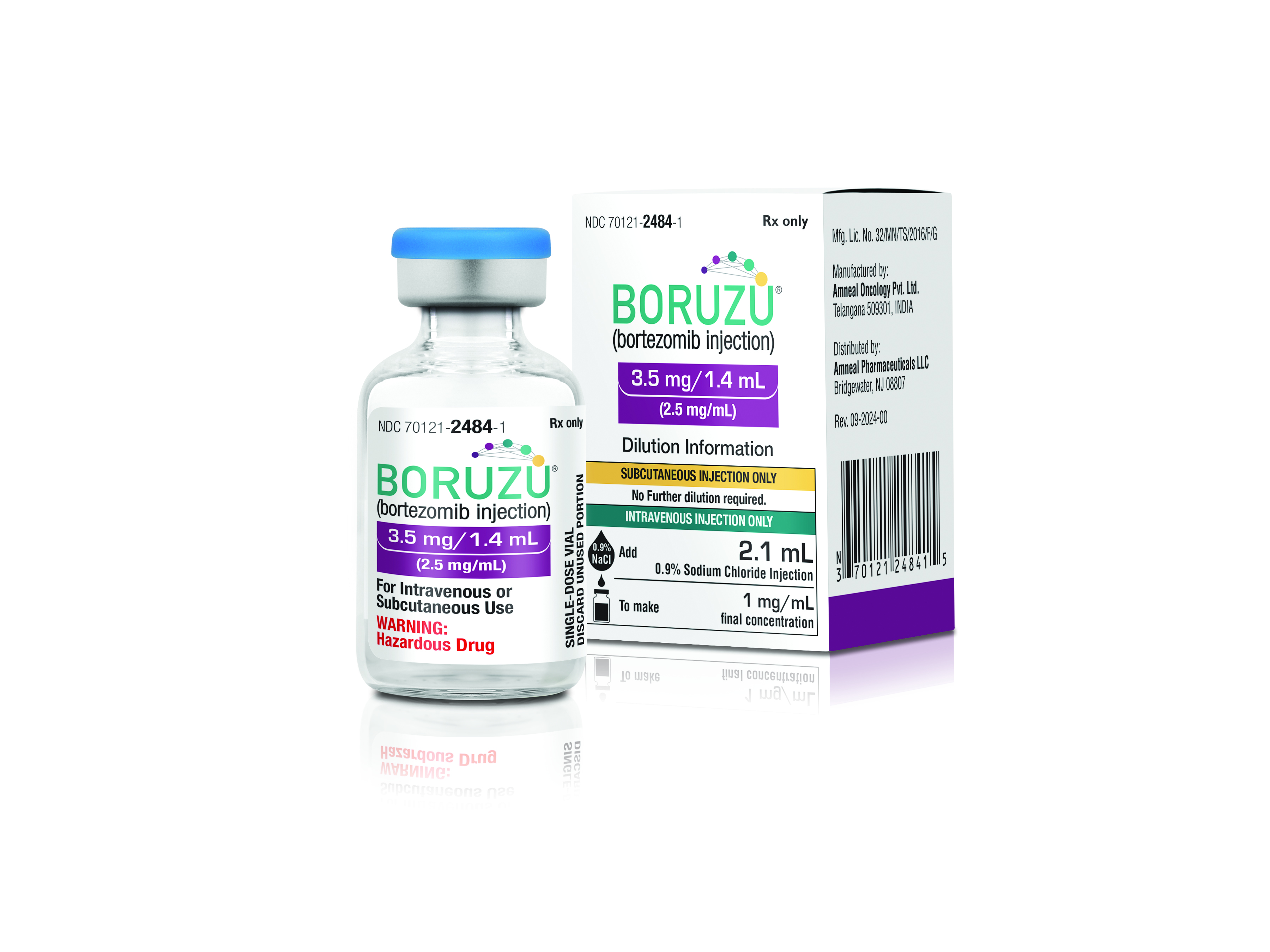
Boruzu (bortezomib injection), a proteasome inhibitor, is used for the treatment of multiple myeloma and mantle cell lymphoma. This product references the branded product Velcade (Takeda), a lyophilized powder requiring reconstitution before use. Shilpa Medicare developed the molecule and Amneal will manufacture and commercialize the product. Boruzu has a unique J-code.
“This ready-to-use injectable marks a significant innovation for our customers by streamlining pharmacy preparation steps for clinicians while now carrying a unique J-code to facilitate reimbursement. Our commitment remains steadfast in delivering differentiated oncology products that enhance patient care and access,” said Sean McGowan, the senior vice president of biosimilars and branded oncology.
“This second NDA [New Drug Application] product being launched in the U.S. market from our novel injectable portfolio demonstrates our capabilities and commitment to introduce pharmacy-efficient solutions that enhance compliance and have the potential to reduce patient wait times,” said Vishnukant Bhutada, the managing director of Shilpa Medicare.
The most commonly reported adverse reactions for Boruzu in clinical studies include asthenic conditions, diarrhea, nausea, constipation, peripheral neuropathy, vomiting, pyrexia, thrombocytopenia, psychiatric disorders, anorexia and decreased appetite, neutropenia, neuralgia, leukopenia, and anemia.
For full prescribing information, see the package insert located here.