Suzetrigine (Journavx, Vertex Pharmaceuticals), a new, nonopioid entry in the treatment armamentarium for moderate to severe acute pain in adults, has garnered considerable attention among clinicians who manage these often challenging patients.
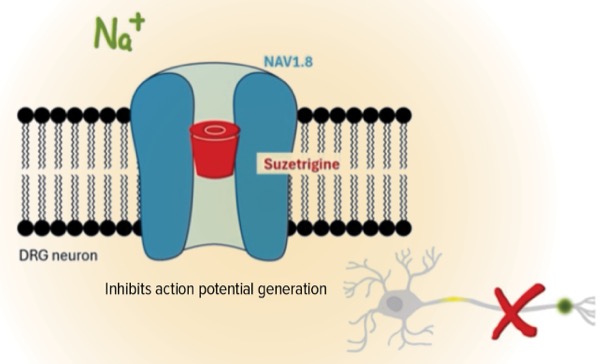
The FDA approved suzetrigine in January as a 50-mg oral tablet. The first-in-class, nonopioid analgesic targets sodium channel pain-signaling pathways in the peripheral nervous system, which prevents pain signals from reaching the brain. The recommended starting dose is 100 mg taken on an empty stomach, and then 50 mg every 12 hours, with or without food.
Suzetrigine’s mechanism of action “is what makes people really excited about this drug, in terms of its selectivity,” said Paige Mathew, PharmD, BCPS, BCGP, a pain management pharmacist with the Veterans Administration and the chair of the Board of Pharmacy Specialties (BPS) Pain Management Specialty Council. “Presumably we are not going to have the concerning central nervous system side effects we see with opioids.”
A person who has experienced opioid use disorder firsthand by seeing it affect family members or their community may hesitate to undergo needed surgery for fear of becoming dependent on opioids. “Now we have something else to [offer these patients],” Dr. Mathew said, adding that she also might refer them to a pain psychologist, social worker or physical therapist for further counseling. During those sessions, the patient’s general concerns before the surgery can be addressed, and they also can be told about the support that will be made available to them afterward, she noted.
Dr. Mathew also encouraged the use of multidisciplinary pain management teams, which she believes are essential because pain is individualized and colored by personal history. She also stressed that offering pain medications without taking a patient’s concerns and perceptions into account might provoke mistrust or lack of adherence, for both acute and chronic pain. “Oftentimes we only focus on the medications,” she said. “We have to consider the biopsychosocial aspects of treating pain as well.”
Key Approval Data
The pivotal trials that led to the FDA approval compared suzetrigine with placebo and the opioid hydrocodone bitartrate/acetaminophen (HB/APAP) for 48 hours following an abdominoplasty or bunionectomy. The abdominoplasty (n=1,118) and bunionectomy (n=1,073) patients had moderate to severe pain following their procedures, as measured by an 11-point Numeric Pain Rating Scale. Self-reports of 6 to 10 on this scale equate to moderate to severe pain.
Researchers tracked reductions in pain scores for patients receiving suzetrigine, placebo or HB/APAP. After 48 hours, participants who had taken suzetrigine had a least squares (LS) mean change from baseline in pain of 118.4, compared with 70.1 for placebo-treated patients and 111.8 for those given HB/APAP (P<0.001). (A larger LS value indicates better efficacy as measured by the time-weighted sum of the pain intensity difference from 0 to 48 hours.) Similar results were reported in a second trial that analyzed suzetrigine for pain reduction in patients undergoing bunionectomies.
The efficacy data are encouraging, noted Mark Garofoli, PharmD, MBA, BCGP, CPE, CTTS, a clinical pain and addiction pharmacist at the West Virginia University Integrative Pain Management Center, in Morgantown. “We need as many options as possible for our patients in pain,” including the judicious use of opioids to manage chronic pain, said Dr. Garofoli, who also serves on the BPS Pain Management Specialty Council.
As for the attention garnered by the suzetrigine approval, it is warranted, Dr. Garofoli said. “This medication has caught more headlines than anyone in the pain realm would have anticipated, and rightfully so,” he said. Like Dr. Mathew, Dr. Garofoli is excited by the opportunity to offer a nonaddictive acute pain medication to his patients.
Given the ongoing scars of the opioid crisis, Dr. Garofoli believes that suzetrigine could sometimes be a hard sell to providers or patients, despite its nonopioid formulation. “People see pain medication and they immediately think addiction,” he said.
But he stressed that opioids will always have some role in pain management. During his career, Dr. Garofoli has seen the pendulum swing between what he perceived as overreliance on opioids and an excessive fear of them. For now at least, suzetrigine is only indicated for acute pain, whereas for chronic conditions, low- to moderate-risk opioid dosages are sometimes still necessary, he noted. “We need to use all the tools in our toolbox as appropriate.”
Anesthesiologist Sees Promise And Challenges
Although suzetrigine inhibits the flow of pain signals via its effects on the NaV1.8 voltage-gated sodium channel, its effectiveness could be limited because other sodium channels (NaV1.1 through 1.9) also transmit pain signals, noted Steven P. Cohen, MD, the division chief of pain medicine at the Northwestern Feinberg School of Medicine, in Chicago. “There will be a ceiling effect,” said Dr. Cohen, who is also the president of the American Society of Regional Anesthesia and Pain Medicine.
In a recent editorial in BMJ Medicine (2025;4[1]:e001431), Dr. Cohen stressed that the true safety risks of suzetrigine will only become known over time. The FDA saw no untoward safety risks that prevented suzetrigine’s approval, but the history of pain medicine includes drugs that were thought to be safer than they were, he noted. For example, in his editorial, Dr. Cohen pointed out that celecoxib (Celebrex, Pfizer) was required to add a black box warning for cardiovascular and gastrointestinal risks soon after its 1998 approval.
Still, Dr. Cohen said he sees promise for suzetrigine, given its nonaddictive mechanism of action. He is a retired colonel, with an interest in lowering the addictive risks of pain medications for military personnel. Suzetrigine can aid in that effort, he said.
The sources reported no relevant financial disclosures.
This article is from the June 2025 print issue.