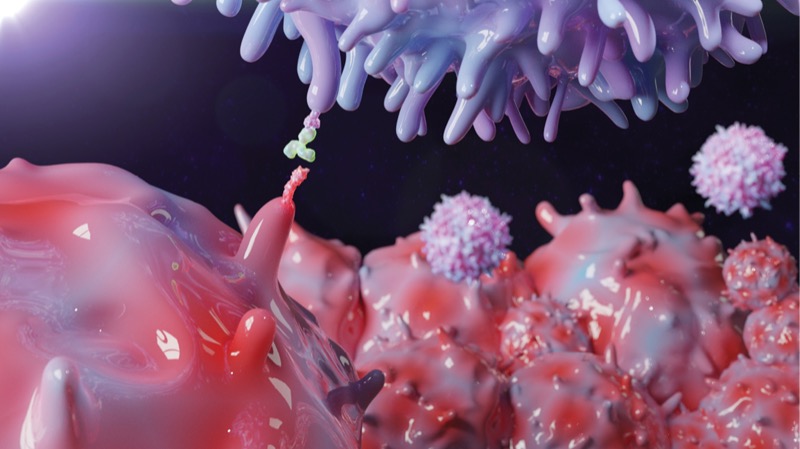
Bispecific antibodies (BsAbs) initially showed promise as an immunotherapy option against hematologic cancers but are now becoming valuable treatments for solid tumors as well, experts said at the 2025 ASCO Annual Meeting, in Chicago. The speakers highlighted the BsAb tarlatamab-dlle (Imdelltra, Amgen), which received FDA approval in 2024 for treating small cell lung cancer (SCLC) that has progressed on or after chemotherapy.
BsAbs target multiple antigens to prime the immune system to fight cancer; tarlatamab-dlle does so by engaging T cells. However, this targeting can provoke excessive immune responses, typically cytokine release syndrome (CRS) or immune effector cell–associated neurotoxicity syndrome (ICANS). Pharmacists with expertise in CRS and ICANS are valuable partners in managing these conditions.
The experience of physicians who specialize in hematologic cancers can also be helpful, because solid cancer clinicians manage CRS and ICANS, ASCO speakers said, particularly hematologists who also work with chimeric antigen receptor T-cell (CAR-T) therapy.
“Heme cancers have had exposure to bispecific T-cell engagers for a longer time, especially in myeloma. And many hematologists also deal with CAR-T cells, which can lead to CRS or ICANS. They have learned to manage and triage those patients appropriately,” said Rafeh Naqash, MD, the director of immuno-oncology at Oklahoma University’s Stephenson Cancer Center, in Oklahoma City.
Generally, tocilizumab is an effective treatment for CRS, and dexamethasone can relieve ICANS. But Dr. Naqash noted that starting such treatments depends on the severity of CRS or ICANS, and requires close monitoring for any adverse reactions.
The initial FDA approval for tarlatamab-dlle in SCLC was accelerated, but Dr. Naqash said he believes full approval will likely soon follow. This is due to positive results of the DeLLphi-304 trial (ClinicalTrials.gov Identifier: NCT05740566) presented at the ASCO meeting; that trial compared tarlatamab-dlle with chemotherapy as a second-line treatment. “DeLLphi-304 showed an overall survival benefit for tarlatamab [J Clin Oncol 2025;43(17 suppl):LBA8008],” Dr. Naqash said. “Previously second-line small cell treatment was chemotherapy, but everyone’s shying away from that and using tarlatamab.”
Dr. Naqash added that tarlatamab-dlle will likely become a first-line therapy for SCLC, given preliminary results from ongoing clinical trials suggesting positive overall survival of tarlatamab-dlle when combined with chemotherapy in the first line. “Small cell lung cancer responds very well to chemotherapy. I don’t think we’ll see chemotherapy taken off the table in the first-line setting,” he said. But the story is different in the second line, he added; here tarlatamab-dlle has already displaced chemotherapy, unless a patient does not respond to it.
Because BsAbs are a new option in SCLC treatment, it may take time for a full picture of their potential side effects to emerge. “The amount of experience we have with tarlatamab is not thousands of patients,” Dr. Naqash said, so it’s possible that some will have delayed-onset CRS or ICANS. This is reminiscent of immune checkpoint inhibitors, he noted, which occasionally cause conditions such as myocarditis.
Tarlatamab-dlle works by targeting the DLL3 protein, which is highly expressed on the surface of cells with SCLC. Other bispecifics that also target DLL3 are in development, Dr. Naqash added.
Focusing on the Patient
The benefits of tarlatamab-dlle for treating SCLC are beyond dispute, noted Melinda Hsu, MD, a thoracic cancer specialist at University Hospitals Seidman Cancer Center, in Cleveland. But clinicians should not discount the real side effects of tarlatamab-dlle and other bispecific T-cell engagers, she said.
The CRS symptoms linked to T-cell engagers range from mild flu-like symptoms to potentially life-threatening inflammation, while ICANS symptoms include seizures and aphasia of varying levels of severity, she noted.
“With efficacy as a given, we’ve all had patients who pepper us with questions,” Dr. Hsu said, adding that patients may ask whether they can go on a cruise or see their grandchildren, or whether their hair will fall out. “What patients are really asking us is, ‘How will this affect my life?’”
Patient-reported outcomes suggest that the BsAb amivantamab-vmjw (Rybrevant, Johnson & Johnson) may have more tolerable side effects than other treatments. In the PAPILLON trial (NCT04538664), a combination of amivantamab-vmjw and chemotherapy demonstrated superior progression-free survival (PFS) than chemotherapy alone (median PFS, 11.4 vs. 6.7 months; P<0.001) (N Engl J Med 2023;389[22]:2039-2051).People who received the combination also reported higher levels of emotional functioning after six months of treatment than those who received chemotherapy alone (63% vs. 47%; P=0.01). Amivantamab-vmjw similarly outperformed a tyrosine kinase inhibitor in the MARIPOSA trial (NCT04487080; J Thorac Oncol 2024;19[10 suppl]:S103-S104).
Although some level of subjectivity is inherent in any self-report, Dr. Hsu said the structured data collection behind these patient reports strengthens their reliability.
Immunotherapies such as BsAbs are among the reasons people with cancer are living longer than they did two decades ago—a development Dr. Hsu considers a great success. But that long-term survival does mean that quality of life is increasingly important; Dr. Hsu noted that some people with metastatic lung cancer may spend much of their time in a clinic or hospital (J Clin Oncol 2022;40[15]:1611-1615). Those prolonged stays could lead to financial stresses, even if someone is very grateful for their increased lifespan.
“There is discrete financial toxicity that exists throughout the cancer journey. I don’t think we have a lot of interventions for that,” Dr. Hsu acknowledged in a post-meeting interview.
Dr. Hsu reported financial relationships with Amgen, AstraZeneca, the Bristol Myers Squibb Foundation, Mirati Therapeutics, MJH Life Sciences, and Regeneron. Dr. Naqash reported financial relationships with Astellas Pharma, Foundation Me, and NGM Biopharmaceuticals.
This article is from the December 2025 print issue.