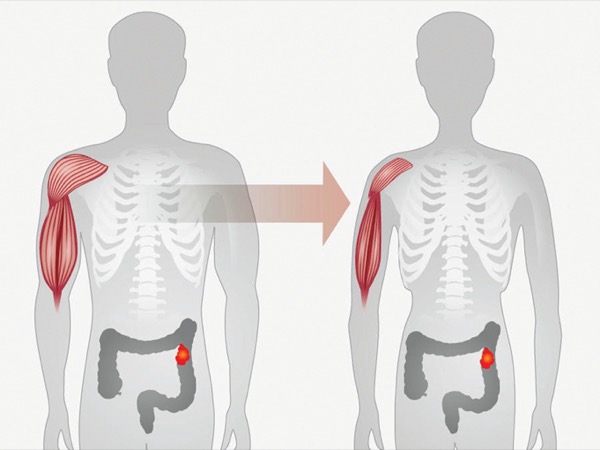
Approximately 80% of patients with advanced cancer experience cachexia, with a one-year mortality rate of 20% to 60%. However, there are no FDA-approved medications to treat the condition. In a session about facing challenging nutritional scenarios in oncology patients, held at the ASPEN 2025 Nutrition Science & Practice Conference, in Columbus, Ohio, experts outlined available treatments for the wasting syndrome.
As cachexia continues to progress, it impedes patients’ ability to tolerate their chemotherapy regimens, noted Anne M. Tucker, PharmD, BCNSP, FASPEN, a clinical pharmacy specialist in critical care/nutrition support at The University of Texas MD Anderson Cancer Center, in Houston. This intolerance can lead to delayed or discontinued chemotherapy, which “contributes to lower survival rates for our patients with cachexia,” Dr. Tucker said, citing published data (J Cachexia Sarcopenia Muscle 2014;5[4]:261-263). She noted these delays also can cause an increase in postoperative complications, fistulas and difficult wound healing, and the condition “also increases our cost of care and [negatively affects] the patients’ quality of life.”
Despite the lack of FDA-approved treatments for cachexia in cancer patients, several organizations—including the American Society of Clinical Oncology (ASCO J Clin Oncol 2020;38[21]:2438-2453) and the European Society for Medical Oncology (ESMO Open 2021;6[3]:100092)—have released guidelines for treatment. These guidelines include recommendations for off-label use of low-dose olanzapine, corticosteroids and progesterone analogs to promote appetite and weight gain. Dr. Tucker explained how the use/benefit of each medication must be weighed against the risks they pose to each patient.
Olanzapine. Approved by the FDA as an atypical antipsychotic agent, olanzapine is used off-label as an appetite stimulant. “A 2023 study really pushed olanzapine to be put in the category of potentially beneficial agents [for treating cancer cachexia]. And that was [noted] in the 2023 ASCO guidelines,” Dr. Tucker explained. This randomized, double-blind, placebo-controlled study (J Clin Oncol 2023;41[14]:2617-2627) compared adding either placebo or 2.5 mg per day of olanzapine to patients undergoing chemotherapy. A significantly greater proportion of patients taking olanzapine than placebo had appetite improvement and higher scores on the Functional Assessment of Anorexia Cachexia Therapy scale: 43% versus 13% (P<0.001) and 22% versus 4% (P=0.004), respectively. However, these benefits come with the risk for sedation, dyslipidemia, increased risk for diabetes, tardive dyskinesia and neuroleptic malignant syndrome, Dr. Tucker noted—all adverse effects cited in the drug’s prescribing information.
Progesterone analogs. Several of these agents, including megestrol acetate, are approved by the FDA to treat AIDS-associated anorexia or cachexia and advanced breast and endometrial cancers. These analogs can cause rash, edema, venous thromboembolism, adrenal insufficiency, impotence and hypertension, according to a representative drug’s prescribing information. Still, Dr. Tucker encouraged the consideration of this agent to treat cancer-related cachexia in patients who can’t tolerate low-dose olanzapine. But she cautioned that a 2024 meta-analysis found that therapy with progesterone analogs among cancer patients with cachexia did not lead to significant weight gain, except in a subgroup analysis (J Cachexia Sarcopenia Muscle 2024;15[4]:1254-1263). A linear dose–response meta-analysis included in the study found that each increment of megestrol acetate (200 mg per day) caused a modest but significant increase in weight gain (maximum effect, 320 mg per day).
Corticosteroids. This drug class is FDA-approved for various conditions and diseases but not cancer-related cachexia. Due to the risk for hyperglycemia, infections, edema, adrenal suppression, peptic ulcer disease, fat redistribution, depression, anxiety, agitation, osteoporosis, myopathy and glaucoma, Dr. Tucker recommended the use of oral dexamethasone 3 to 4 mg dosed daily (or equivalent) for two to three weeks in patients with cancer-related cachexia who do not tolerate olanzapine and have a short life expectancy.
“Weighing these adverse effects is what guides you in selecting among these treatments and how they fit in with your patients’ other comorbidities,” Dr. Tucker said. However, she pointed out that the evidence supporting their use for cancer-related cachexia is insufficient to lead to FDA approval.
Asked to comment on these three classes of drugs, Todd Mattox, PharmD, RPh, BCNSP, FASPEN, a medicine/surgery clinical pharmacist at Moffitt Cancer Center, in Tampa, Fla., noted that, based on published data, they “appear to provide the best chance of producing positive results on appetite.”
Dr. Mattox added that the reported weight gain from these agents “has been extremely variable to nonexistent, but some would argue that a clinical success for many patients is to stop or slow weight loss. The choice of which agent to use is patient-situation dependent, based on underlying comorbid conditions, adverse effect profile and the patient’s expected life expectancy.”
Cachexia Pipeline Agent
The cancer-related cachexia pipeline is robust, Dr. Tucker noted. One promising compound is ponsegromab (Pfizer), a humanized monoclonal antibody that inhibits growth differentiation factor, which is a cytokine elevated in cancer-related cachexia. Dr. Tucker said she was “excited” about a phase 2 study of 187 participants with non-small cell lung cancer, pancreatic cancer or colorectal cancer, in which ponsegromab therapy yielded significant increases in body weight after 12 weeks across all doses: 2.02% (95% CI, –0.97% to 5.01%) in a 100-mg treatment group, 3.48% (95% CI, 0.54%-6.42%) in a 200-mg group and 5.61% (95% CI, 2.56%-8.67%) in a 400-mg group, compared with placebo, according to data announced by Pfizer. No clinically significant adverse trends were noted with ponsegromab administration.
Dr. Mattox reported no relevant financial disclosures. Dr. Tucker is a consultant to UpToDate Inc.
This article is from the May 2025 print issue.