A new review of all 50 state boards of pharmacy regulations by the Institute for Safe Medication Practices has shown that only one or two boards provided detailed guidance for pharmacy drug labeling, ISMP presenters noted at the NASP 2024 Annual Meeting & Expo, in Nashville, Tenn.
“Most boards have just generalized, high-level suggestions outside listing requirements for a legal label,” said Jennifer Young, PharmD, BCPS, CSP, a medication safety specialist with ISMP, who noted that an ISMP fellow prepared the pharmacy board review in preparation for the NASP presentation. “I’ve worked in a variety of places in my career, and in each pharmacy, sometimes each store, they had their own preferences and nuances.”
The National Association of Boards of Pharmacy published the findings of a task force on prescription container labeling in 2009, but Dr. Young noted that the findings mostly focus on the patient-facing prescription label, stating that critical information must appear on the label with emphasis in a sans serif, minimum 12-point font in “sentence case” that contains numeric rather than alphabetic characters and simplified language that avoids unfamiliar words and medical jargon.
USP General Chapter <17> on prescription container labeling, most recently updated in 2021, includes more detail, she said. “It recommends the use of explicit instructions, including standardized time periods (morning, noon, evening, bedtime) or with lunch, at dinner, rather than ‘take one tablet twice daily,’ along with similar recommendations to avoid alphanumeric characters for numbers and use familiar language rather than medical jargon.”
Of the state boards, California’s is by far the most detailed in its recommendations, Dr. Young said. “In a 2024 update to their Lawbook for Pharmacy, they specifically call out how the prescription language should be written. For example, they stipulate that it should state, ‘Take x dosage of y form,’ not strength. They also list examples of how instructions should be written, such as ‘take 1 [insert appropriate dosage form] at bedtime.’” New Jersey’s administrative code allows pharmacists to make minor changes to prescription labeling to ensure the prescribing intent is clear to the patient, she added.
Dr. Young recommended that specialty pharmacies make use of ISMP’s 2023-2024 Targeted Medication Safety Best Practices for Community Pharmacy. “Best practice number 4 states that pharmacies should standardize the use of metric units of measure when prescribing, dispensing and measuring oral liquid medications,” she noted. ISMP also offers a List of Error-Prone Abbreviations, Symbols, and Dose Designations, which also standardizes the use of metric units, demonstrates how to safely express decimals and fractions of dose units, and suggests how to avoid other error-prone abbreviations, symbols and dose designations.
ISMP recently added a new medication resource center to its medication safety membership, which includes guidance documents focused on outlining the anatomy of the prescription label, principles for creating patient-centered prescription directions and a curated literature search.
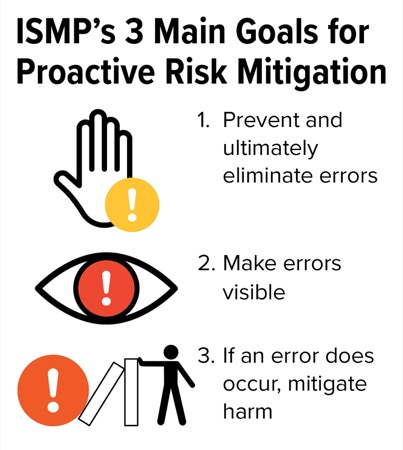
3 Primary Goals
As for more generalized medication safety tips, specialty pharmacies should combine low-, medium- and high-leverage risk-reduction strategies to prevent medication errors, Dr. Young noted. “Healthcare is complex, and risk is everywhere,” she said. “Keep in mind three primary goals for medication safety when working toward proactively controlling risk: We want to prevent and ultimately eliminate errors; we want to make errors visible; and if an error does occur, we want to mitigate harm. The intent is to consider all three of these goals when choosing effective strategies to reduce risk.”
ISMP has published a hierarchy of risk mitigation strategies, ranging from high leverage (typically the most effective but hardest to implement) to low leverage (least effective, but easier to implement). “The top, high-leverage strategies rely on systems, and are the most effective because they eliminate the risk for errors and associated harm by designing out hazards through interventions like forcing functions, barriers and fail-safes, and automation. However, they often require complex implementation plans,” Dr. Young said.
Medium-leverage strategies such as standardized protocols, warnings and checklists reduce the likelihood of errors or minimize harm, but may need periodic updating and reinforcement. Low-leverage, easy-to-implement strategies such as educational programs and rules and policies, while important, are insufficient, she noted.
“To be clear, education absolutely has its place as a prerequisite, providing teammates with the required knowledge and skills to do their job well,” she said. “Education is often necessary to review new drugs hitting the market, new medication delivery devices, and new or changed automation processes.” However, “education must be coupled with a medium- and/or high-level strategy to drive the greatest impact on system reliability and patient safety.”
Better Benchmarking
After addressing these overarching medication strategies, more specific risks can be tackled, the ISMP presenters noted. One particularly challenging safety issue is the misuse of medication error rate data, said Christina Michalek, BS, RPh, the director of ISMP’s Membership and Patient Safety Organization—a concern illustrated by queries that ISMP has received about medication error rates over the years (sidebar). ISMP defines benchmarking as “an ongoing process that determines how other organizations have achieved the best performance and suggests ways for adapting the best practices that result in this exceptional performance.”
For benchmarking to be effective, “it’s got to be a dual process,” Ms. Michalek said. “You need to measure performance, but you also need to know the practices that are going to lead to that performance. We question the wisdom of applying a benchmark of error rates to the medication-use process because the true incidence of errors varies for a number of different reasons, and people can fall into the mistaken belief that the benchmark is simply about comparing the numbers and not identifying the enablers of good performance.”
Many organizations, including accreditation bodies and third-party payors, require collection and tracking of medication error data. “They are looking for lower rates, representing better performance,” Ms. Michalek noted. “Some payors require pharmacies maintain a specific accuracy rate; if an error rate is too high, the payor contract may be lost,” she said. “But with error reporting, we argue that less is definitely not more. There really is no consistent way to detect and report errors, and we know that many errors go undetected. So, if you have a high error rate, does that mean that you really have unsafe practices, or might it mean that you have a really healthy reporting culture? If you have a low error rate, does that mean that you’re very successful at preventing errors? Or maybe your culture is punitive?”
ISMP and the National Coordinating Council for Medication Error Reporting and Prevention do not want to see reporting rates for medication errors or adverse drug events used as a means for internal or external comparison. “Instead, what is more important is the quality of the information collected, data analysis and system improvements to prevent patient harm,” Dr. Michalek said. “What do you do when you learn about a risk? What are the process improvements that you put in place when you have a risk identified? We recommend that you strive for increased actionable reporting, using your reporting systems to generate impactful changes and communicate those with the frontline staff so that they give you more and more information and you become more proactive about risk reduction.”
More useful ways to collect data, she said, include:
- technology such as medication barcode scanning used during fulfillment;
- culture of safety surveys such as the Agency for Healthcare Research and Quality’s Surveys on Patient Safety Culture; and
- data to quantify system changes.
“You can build an interactive dashboard with a number of these things together,” she said. “If you are required by an organization to collect and report an error rate, we will tell you that there is no national comparison, and we would rather see organizations look for a demonstration of movement toward safer and more reliable medication-use systems.”
The sources reported no relevant financial disclosures.
This article is from the February 2025 print issue.