Department of Pharmacy Practice
University of Illinois Chicago College of Pharmacy
Department of Pharmacy Practice
University of Illinois
Chicago College of Pharmacy
Chronic lymphocytic leukemia (CLL) is a lymphoproliferative disorder characterized by the accumulation of circulating mature B cells that are functionally incompetent and usually monoclonal in origin.1 Small lymphocytic lymphoma (SLL) refers to lymphadenopathy and/or extranodal involvement of monoclonal B cells with a CLL immunophenotype, with relatively few or no circulating malignant lymphocytes; thus, CLL and SLL are different manifestations of the same underlying disease process. CLL/SLL primarily affects older adults.
The CLL/SLL treatment landscape evolved significantly with the introduction of targeted therapies, including covalent Bruton’s tyrosine kinase (BTK) inhibitors and the B-cell lymphoma 2 (BCL2) inhibitor venetoclax (Venclexta, AbbVie), in the 2010s.2-4 Treatment remains challenging given the lack of consensus on sequencing therapies and variability among treatments in their supporting evidence and toxicities. While the non-covalent BTK inhibitor pirtobrutinib (Jaypirca, Eli Lilly) and chimeric antigen receptor (CAR) T-cell therapies have roles after failure of covalent BTK inhibitors and venetoclax, there are insufficient data to guide therapy in patients who are ineligible for these later-line therapies or in whom they have failed, necessitating an individualized approach to treatment.5
Epidemiology
CLL/SLL is among the most common hematologic malignancies in developed nations, accounting for approximately one-third of all leukemia diagnoses in the United States.6 SLL constitutes approximately 15% of CLL/SLL cases.7 Globally, the age-adjusted incidence of CLL/SLL is approximately 4 to 5 cases per 100,000 population.1 The disease is more common in men; in the United States, incidence rates among men and women are estimated at 6.75 and 3.65 cases per 100,000 population per year, respectively.8 The median age at diagnosis of CLL/SLL is approximately 70 years, and less than 2% of affected patients are younger than 45 years.8,9 CLL/SLL is more commonly diagnosed in White individuals than in Hispanic, Black, and Asian patients.2
Clinical Presentation
Most commonly, CLL/SLL is diagnosed incidentally when a routine complete blood count indicates absolute lymphocytosis; most patients are asymptomatic at this time.2,10 The second most common presentation involves a painless lymphadenopathy, often in the cervical area. Less frequently, patients may present with “B symptoms” of lymphoma, a term used to denote the presence of fever, fatigue, drenching night sweats, and the unintended loss of more than 10% of body weight over 6 months.1 Cytopenias also may be present due to marrow infiltration.
Staging and Prognosis
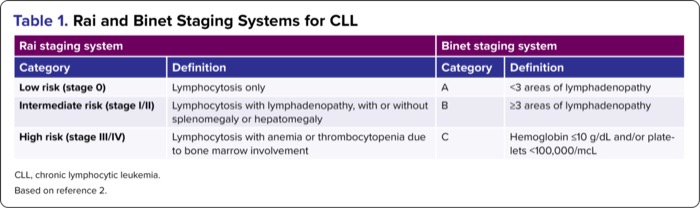
The Rai and Binet staging systems are widely used to stage CLL; these systems are compared in Table 1.2 The Rai system is more commonly used in the United States, while the Binet system is more common in Europe. Both systems categorize patients into stages of disease reflecting predicted overall survival (OS). Although still used for their prognostic utility and to aid in treatment decision-making, OS has improved dramatically in patients with CLL since the introduction of the Rai and Binet systems in the 1970s and 1980s, making the systems’ generalizability to current practice somewhat limited.11 SLL is staged radiographically using the Lugano modification of the Ann Arbor staging system for B-cell non-Hodgkin lymphomas.12
| Table 1. Rai and Binet Staging Systems for CLL | |||
| Rai staging system | Binet staging system | ||
|---|---|---|---|
| Category | Definition | Category | Definition |
| Low risk (stage 0) | Lymphocytosis only | A | <3 areas of lymphadenopathy |
| Intermediate risk (stage I/II) | Lymphocytosis with lymphadenopathy, with or without splenomegaly or hepatomegaly | B | =3 areas of lymphadenopathy |
| High risk (stage III/IV) | Lymphocytosis with anemia or thrombocytopenia due to bone marrow involvement | C | Hemoglobin =10 g/dL and/or platelets <100,000/mcL |
| CLL, chronic lymphocytic leukemia. Based on reference 2. | |||
Although it is not widely used, the CLL-International Prognostic Index (CLL-IPI) is a more recently proposed prognostic scoring system that integrates major clinical, biochemical, and genetic prognostic markers for patients with CLL/SLL.2,13 Five-year OS predictions using CLL-IPI range from approximately 93% in low-risk patients to 23% in very high-risk patients; however, because CLL-IPI was developed and validated in patients receiving front-line chemoimmunotherapy (CIT), these risk scores may also underestimate survival.14
Predictors of Treatment Outcome
Specific markers have been identified that predict the likelihood of favorable treatment outcome in CLL/SLL and, therefore, can influence treatment selection.15 Among these, alterations to the TP53 tumor suppressor and IGHV genes are assessed routinely during pretreatment evaluation. TP53 abnormalities (ie, del[17p] or TP53 mutations) have been associated with poorer clinical response to chemotherapy and CIT.11 IGHV encodes part of the B-cell receptor, and unmutated status typically predicts more aggressive disease and shorter survival after CIT. In contrast, unmutated IGHV has been demonstrated in clinical trials to be predictive of benefit of regimens containing both venetoclax and obinutuzumab (Gazyva, Genentech) over CIT, although patients with unmutated IGHV experience worse outcomes than those with mutated IGHV receiving the same treatment.16-18 Although IGHV status does not change over time, TP53 abnormalities may be acquired; thus, TP53 status typically is reassessed at each new line of therapy. Complex karyotypes (with the notable exception of cases with trisomies of both chromosomes 12 and 19) portend a poorer CIT prognosis, and recent data indicate that this association also extends to patients treated with front-line venetoclax-based regimens.19,20
Management
Several principles guide the management of CLL/SLL.11,21 Because CLL and SLL represent the same underlying disease, nearly all cases follow identical management pathways. Most cases present without symptoms, are diagnosed after incidental findings, and can therefore be observed without immediate treatment.10 There is no known OS benefit associated with treating observable CLL/SLL; thus, outside of clinical trials, patients with CLL/SLL, including those who have received prior therapy, should have an indication for treatment (Table 2).22,23 The goals of CLL/SLL management are palliative for most patients: When treatment is indicated, conventional systemic therapy prolongs life and relieves symptoms but does not result in a cure. Although CLL/SLL can be cured with allogeneic hematopoietic cell transplantation, this approach is associated with significant morbidity and mortality, and is reserved for highly selected cases.
| Table 2. Indications for Treatment of CLL/SLL |
| Autoimmune complications (eg, steroid-refractory cytopenias) |
| Bulky (=6 cm below costal margin) or symptomatic splenomegaly |
| Bulky (=10 cm) or symptomatic lymphadenopathy |
| Extranodal disease–related symptoms or threatened end-organ function |
| Progressive lymphocytosis (eg, lymphocyte doubling time <6 mo)a |
| Progressive anemia (eg, hemoglobin <10 g/dL) |
| Progressive thrombocytopenia (eg, platelets <100×109/L) |
Other disease-related symptoms:
|
| CLL, chronic lymphocytic leukemia; SLL, small lymphocytic lymphoma. a Elevated absolute lymphocyte count alone is not an unequivocal indication for treatment, as leukostatic complications are exceedingly rare in CLL/SLL. Based on references 21 and 24. |
Treatment Overview
Although a small proportion of patients with SLL present with localized disease, which some experts may treat with radiotherapy alone, most patients with CLL/SLL who require treatment receive systemic therapy.10,11,21,25-27 Until recently, standard initial treatment was CD20 monoclonal antibody–based CIT.28 Superior survival outcomes with covalent BTK inhibitors and venetoclax have supplanted the use of CIT, which now is largely relegated to selected circumstances (eg, BTK inhibitors and venetoclax are inaccessible; multiple failed prior therapies).10,11
Because covalent BTK inhibitor– and venetoclax-based therapies have not yet been directly compared as initial treatment, differentiating factors for selecting among these agents in the front-line setting include key drug characteristics (outlined below) and other aspects of shared decision-making (eg, patient preference for continuous treatment with a covalent BTK inhibitor vs fixed-duration treatment with venetoclax and obinutuzumab). Mounting clinical experience with acalabrutinib (Calquence, AstraZeneca) and zanubrutinib (Brukinsa, BeiGene) has led to their preferential use over ibrutinib (Imbruvica, Pharmacyclics) in the front-line setting based on improved tolerability.10,29 Additionally, in patients with del(17p) or TP53 mutations, some experts preferentially recommend covalent BTK inhibitor–based regimens or, alternatively, continuous venetoclax-based regimens as initial therapy over fixed-duration venetoclax-based treatment; these strategies are based on encouraging outcomes in front-line covalent BTK inhibitor trials, subgroup analysis suggesting unfavorable outcomes with fixed-duration venetoclax-based treatment, and extrapolation of findings with long-term or continuous venetoclax dosing in the relapsed/refractory setting.10
These factors also drive decisions for patients requiring second- and subsequent-line treatment, with the added consideration that such choices must incorporate treatment history.10,11 For example, in patients previously treated with BTK inhibitor(s) or venetoclax who experienced disease progression during therapy, treatment with an agent from the opposite class should be considered; in those who experienced clinical benefit with prior covalent BTK inhibitor therapy and discontinued treatment due to toxicity, treatment with an alternative covalent BTK inhibitor or venetoclax should be considered.
Consideration also can be given to repeating a regimen in patients who experienced clinical benefit with prior fixed-duration therapy. In patients whose prior treatment has not included a covalent BTK inhibitor, head-to-head comparisons have indicated that, relative to ibrutinib, zanubrutinib is associated with superior progression-free survival (PFS), and both acalabrutinib and zanubrutinib have improved safety profiles.10 The non-covalent BTK inhibitor pirtobrutinib has demonstrated efficacy in patients with covalent BTK inhibitor–resistant disease and should be considered after treatment with covalent BTK inhibitors and venetoclax has failed.30,31 CD19-directed CAR T-cell therapy with lisocabtagene maraleucel (Breyanzi, Bristol Myers Squibb) is also an established option for eligible patients after BTK inhibitor– and venetoclax-based treatment.32,33
Front-Line Treatment With Covalent BTK Inhibitors
BTK inhibitors disrupt B-cell receptor signal transduction, impairing aberrant survival pathways and promoting cell death in malignant B lymphocytes.34 The first-generation, first-in-class BTK inhibitor ibrutinib and second-generation BTK inhibitors acalabrutinib and zanubrutinib are FDA approved for the treatment of CLL/SLL.35-38 These agents bind irreversibly to BTK and are referred to as covalent BTK inhibitors. Several major trials, outlined below, established preferential use of the covalent BTK inhibitors as initial monotherapy—or, in the case of acalabrutinib, in combination with obinutuzumab—over CIT in patients with CLL/SLL.
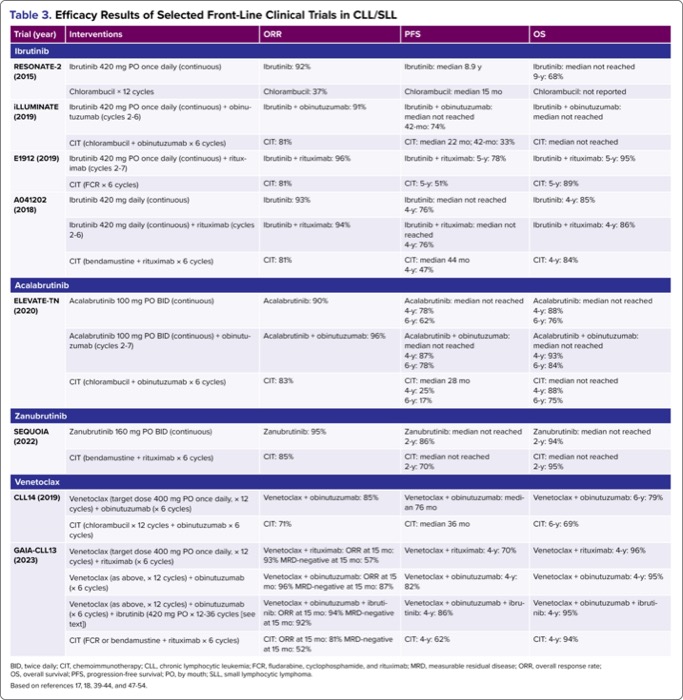
Ibrutinib’s approval for front-line treatment of CLL/SLL was based on improved survival compared with chlorambucil in older patients enrolled in the randomized, open-label, phase 3 RESONATE-2 trial (Table 3).39,40 Of note, after RESONATE-2 had been initiated, the CLL11 trial demonstrated that CIT with chlorambucil plus obinutuzumab produced superior survival outcomes to chlorambucil alone; as a result of these trials, chlorambucil in combination with a CD20 monoclonal antibody was previously considered a standard-of-care front-line treatment option alongside ibrutinib monotherapy for patients unfit for more aggressive CIT regimens.1 The randomized, open-label, phase 3 iLLUMINATE trial compared ibrutinib and chlorambucil, both in combination with obinutuzumab, in this population with superior PFS in the ibrutinib-based arm.41,42 In younger and/or more fit patients with previously untreated CLL/SLL, superior survival with ibrutinib-based therapy versus standard-of-care CIT was established in the randomized, open-label, phase 3 E1912 trial comparing ibrutinib plus rituximab with fludarabine, cyclophosphamide, and rituximab.43,44 A propensity score–matched emulated comparison using patient-level data supported an OS benefit associated with initial treatment with ibrutinib over initial treatment with CIT followed by second-line ibrutinib.45 Similar findings were reported in an analysis of prospective registry data, which further supported prolonged time to next line of therapy and reduced rates of adverse events with initial treatment with ibrutinib compared with initial treatment with CIT.46
| Table 3. Efficacy Results of Selected Front-Line Clinical Trials in CLL/SLL | ||||
| Trial (year) | Interventions | ORR | PFS | OS |
|---|---|---|---|---|
| Ibrutinib | ||||
| RESONATE-2 (2015) | Ibrutinib 420 mg PO once daily (continuous) | Ibrutinib: 92% | Ibrutinib: median 8.9 y | Ibrutinib: median not reached 9-y: 68% |
| Chlorambucil × 12 cycles | Chlorambucil: 37% | Chlorambucil: median 15 mo | Chlorambucil: not reported | |
| iLLUMINATE (2019) | Ibrutinib 420 mg PO once daily (continuous) + obinutuzumab (cycles 2-6) | Ibrutinib + obinutuzumab: 91% | Ibrutinib + obinutuzumab: median not reached 42-mo: 74% | Ibrutinib + obinutuzumab: median not reached |
| CIT (chlorambucil + obinutuzumab × 6 cycles) | CIT: 81% | CIT: median 22 mo; 42-mo: 33% | CIT: median not reached | |
| E1912 (2019) | Ibrutinib 420 mg PO once daily (continuous) + rituximab (cycles 2-7) | Ibrutinib + rituximab: 96% | Ibrutinib + rituximab: 5-y: 78% | Ibrutinib + rituximab: 5-y: 95% |
| CIT (FCR × 6 cycles) | CIT: 81% | CIT: 5-y: 51% | CIT: 5-y: 89% | |
| A041202 (2018) | Ibrutinib 420 mg daily (continuous) | Ibrutinib: 93% | Ibrutinib: median not reached 4-y: 76% | Ibrutinib: |
| Ibrutinib 420 mg daily (continuous) + rituximab (cycles 2-6) | Ibrutinib + rituximab: 94% | Ibrutinib + rituximab: median not reached 4-y: 76% | Ibrutinib + rituximab: | |
| CIT (bendamustine + rituximab × 6 cycles) | CIT: 81% | CIT: median 44 mo 4-y: 47% | CIT: | |
| Acalabrutinib | ||||
| ELEVATE-TN (2020) | Acalabrutinib 100 mg PO BID (continuous) | Acalabrutinib: 90% | Acalabrutinib: median not reached 4-y: 78% 6-y: 62% | Acalabrutinib: median not reached 4-y: 88% 6-y: 76% |
| Acalabrutinib 100 mg PO BID (continuous) + obinutuzumab (cycles 2-7) | Acalabrutinib + obinutuzumab: 96% | Acalabrutinib + obinutuzumab: median not reached 4-y: 87% 6-y: 78% | Acalabrutinib + obinutuzumab: median not reached 4-y: 93% 6-y: 84% | |
| CIT (chlorambucil + obinutuzumab × 6 cycles) | CIT: 83% | CIT: median 28 mo 4-y: 25% 6-y: 17% | CIT: median not reached 4-y: 88% 6-y: 75% | |
| Zanubrutinib | ||||
| SEQUOIA (2022) | Zanubrutinib 160 mg PO BID (continuous) | Zanubrutinib: 95% | Zanubrutinib: median not reached 2-y: 86% | Zanubrutinib: median not reached 2-y: 94% |
| CIT (bendamustine + rituximab × 6 cycles) | CIT: 85% | CIT: median not reached 2-y: 70% | CIT: median not reached 2-y: 95% | |
| Venetoclax | ||||
| CLL14 (2019) | Venetoclax (target dose 400 mg PO once daily, × 12 cycles) + obinutuzumab (× 6 cycles) | Venetoclax + obinutuzumab: 85% | Venetoclax + obinutuzumab: median 76 mo | Venetoclax + obinutuzumab: |
| CIT (chlorambucil × 12 cycles + obinutuzumab × 6 cycles) | CIT: 71% | CIT: median 36 mo | CIT: | |
| GAIA-CLL13 (2023) | Venetoclax (target dose 400 mg PO once daily, × 12 cycles) + rituximab (× 6 cycles) | Venetoclax + rituximab: ORR at 15 mo: 93% MRD-negative at 15 mo: 57% | Venetoclax + rituximab: | Venetoclax + rituximab: |
| Venetoclax (as above, × 12 cycles) + obinutuzumab (× 6 cycles) | Venetoclax + obinutuzumab: ORR at 15 mo: 96% MRD-negative at 15 mo: 87% | Venetoclax + obinutuzumab: | Venetoclax + obinutuzumab: | |
| Venetoclax (as above, × 12 cycles) + obinutuzumab (× 6 cycles) + ibrutinib (420 mg PO × 12-36 cycles [see text]) | Venetoclax + obinutuzumab + ibrutinib: ORR at 15 mo: 94% MRD-negative at 15 mo: 92% | Venetoclax + obinutuzumab + ibrutinib: | Venetoclax + obinutuzumab + ibrutinib: | |
| CIT (FCR or bendamustine + rituximab × 6 cycles) | CIT: ORR at 15 mo: 81% MRD-negative at 15 mo: 52% | CIT: | CIT: | |
| BID, twice daily; CIT, chemoimmunotherapy; CLL, chronic lymphocytic leukemia; FCR, fludarabine, cyclophosphamide, and rituximab; MRD, measurable residual disease; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PO, by mouth; SLL, small lymphocytic lymphoma. Based on references 17, 18, 39-44, and 47-54. | ||||
RESONATE-2, iLLUMINATE, and E1912 were designed with the assumption that addition of a CD20 monoclonal antibody improves outcomes over ibrutinib monotherapy, based on prior experience in CIT trials.1,55 This assumption was evaluated in the randomized, phase 3 A041202 trial comparing ibrutinib alone, ibrutinib plus rituximab, and CIT in older patients with previously untreated CLL/SLL.47 Ibrutinib with or without rituximab produced superior PFS to CIT, with no difference in PFS between patients treated with ibrutinib alone and those with ibrutinib plus rituximab. Extended
The second-generation covalent BTK inhibitors were developed, in part, to improve upon ibrutinib’s target affinity and specificity.34 Acalabrutinib with or without obinutuzumab was compared with CIT in previously untreated patients with CLL/SLL in the randomized, open-label, phase 3 ELEVATE-TN trial; similar to the A041202 trial, PFS was significantly prolonged in patients receiving acalabrutinib with or without obinutuzumab relative to those receiving chlorambucil plus obinutuzumab (Table 3).49,50 In extended
As noted previously, patients with del(17p) or TP53 mutations are predisposed to inferior outcomes, including poor response rate and duration when treated with chemotherapy or CIT; this long-recognized phenomenon rendered treatment of this subpopulation particularly difficult in the pre–BTK inhibitor era.56 Ibrutinib was the first treatment approved specifically for patients with CLL/SLL with del(17p).57 This approval was initially based on the phase 3 RESONATE trial (described in the next installment in this series) in patients with relapsed/refractory CLL/SLL, which recruited 127 patients with del(17p); although the study was not conducted in the front-line setting, the approved labeling included patients with del(17p) regardless of treatment history due to impressive results and a lack of other options for this subpopulation.58 Ibrutinib monotherapy was also studied in 34 patients with CLL/SLL with del(17p) or mutated TP53 regardless of treatment status in a single-arm phase 2 trial, and the RESONATE-2, iLLUMINATE, and E1912 trials recruited 11, 18, and 26 patients with del(17p) or mutated TP53, respectively.39,41,43,59 Pooled analysis of these cohorts indicated excellent outcomes and suggested that survival rates may be similar to those without del(17p) or mutated TP53 treated with ibrutinib.60 Similarly encouraging findings were noted in the subsets of patients with del(17p) or mutated TP53 treated with acalabrutinib (n=48) or zanubrutinib (n=110) in the ELEVATE-TN and SEQUOIA trials, respectively.49,51,52
Front-Line Treatment With Venetoclax
Venetoclax inhibits the pro-survival protein BCL2, which is constitutively overexpressed in CLL/SLL cells, thereby facilitating apoptotic cell death.61 The preferential front-line use of venetoclax-based therapy over CIT was established in the randomized, open-label CLL14 trial comparing venetoclax plus obinutuzumab with chlorambucil plus obinutuzumab (Table 3).17 In contrast to the BTK inhibitors, the venetoclax combination was administered for a fixed duration (12 months); patients receiving venetoclax plus obinutuzumab experienced significantly longer PFS than those assigned to CIT. Like other major trials in this setting, CLL14 was not designed to compare outcomes in patients with TP53 abnormalities; however, PFS appeared to be significantly prolonged in patients with del(17p) and/or mutated TP53 who received venetoclax plus obinutuzumab than those who received CIT with extended
The randomized, open-label, phase 3 GAIA-CLL13 trial compared 3 fixed-duration venetoclax-based regimens—venetoclax in combination with rituximab, obinutuzumab, or both obinutuzumab and ibrutinib—with CIT in patients without del(17p) or TP53 abnormalities.18 Uniquely, one of the co-primary end points in this trial was measurable residual disease (MRD) negativity, which guided treatment in the venetoclax-obinutuzumab-ibrutinib arm. Patients in this group who had not achieved MRD-negative response with 12 cycles of treatment continued ibrutinib for 36 cycles. Patients assigned to venetoclax plus obinutuzumab or venetoclax-obinutuzumab-ibrutinib, but not venetoclax plus rituximab, were significantly more likely to have undetectable MRD after 15 months than those who received CIT. In a preplanned interim analysis of PFS (the second co-primary end point) with a median 38.8 months of
Front-Line Treatment With Other Agents
Because front-line treatment with a covalent BTK inhibitor– or venetoclax-based regimen is associated with superior outcomes and these agents are relatively well tolerated, situations in which other front-line regimens must be used are unusual and require an individualized approach.10 In patients who are unable to receive targeted therapy (eg, lack of access or immitigable drug interactions) but are otherwise relatively fit for treatment, CIT may still be considered for those without del(17p) or TP53 mutations.11 In patients unable to receive targeted therapy who are considered unfit for CIT, options are limited; some data indirectly support the use of obinutuzumab monotherapy in this setting.11,62,63
Dr. Buege reported no relevant financial disclosures. Dr. Rodriguez was employed at the University of Illinois Chicago College of Pharmacy at the time of initial writing of this review. He is now employed at Novo Nordisk Inc but disclosed that the initial publication was written in his own capacity, not on behalf of the company. Dr. Rodriguez has not been involved in writing updates to this review since the initial version published in July 2023.
References
- Hallek M, et al. Lancet. 2018;391(10129):1524-1537.
- Woyach JA, et al. In: Loscalzo J, et al, eds. Harrison’s Principles of Internal Medicine. 21st ed. McGraw-Hill Education; 2022.
- Roeker LE, et al. Drugs. 2022;82:133-143.
- FDA. Drugs@FDA. Accessed November 8, 2024. https://www.accessdata.fda.gov/scripts/cder/daf/
- Huang IJ, et al. Expert Opin Pharmacother. Published online September 8, 2024. doi:10.1080/14656566.2024.2398603
- Siegel RL, et al. CA Cancer J Clin. 2024;74(1):12-49.
- Tsimberidou AM, et al. J Clin Oncol. 2007;25:4648-4656.
- Yamamoto JF, et al. Cancer Causes Control. 2008;19(4):379-390.
- Kutsch N, et al. Am J Hematol. 2022;97(suppl 2):S3-S10.
- Shadman M. JAMA. 2023;329(11):918-932.
- Wierda WG, et al. J Natl Compr Canc Netw. 2024;22(3):175-204.
- Cheson BD, et al. J Clin Oncol. 2014;32(27):3059-3068.
- International CLL-IPI working group. Lancet Oncol. 2016;17(6):779-790.
- Gentile M, et al. Eur J Haematol. Published online July 24, 2018. doi:10.1111/ejh.13149
- Cohen JA, et al. Cancers (Basel). 2020;12(4):894.
- Tausch E, et al. Blood. 2020;135(26):2402-2412.
- Fischer K, et al. N Engl J Med. 2019;380(23):2225-2236.
- Eichhorst B, et al. N Engl J Med. 2023;388(19):1739-1754.
- Furstenau M, et al. Blood. 2023;142:446-459.
- Baliakas P, et al. Blood. 2019;133(11):1205-1216.
- Hallek M, et al. Blood. 2018;131(25):2745-2760.
- CLL Trialists’ Collaborative Group. J Natl Cancer Inst. 1999;91(10):861-868.
- Langerbeins P, et al. Blood. 2022;139(2):177-187.
- Singh N, et al. Am J Case Rep. 2020;21:e924798.
- Morrison WH, et al. J Clin Oncol. 1989;7(5):598-606.
- Hoskin PJ, et al. Lancet Oncol. 2014;15(4):457-463.
- Eichhorst B, et al. Ann Oncol. 2021;32(1):23-33.
- Gribben JG. Blood. 2010;115:187-197.
- Hampel PJ, et al. Blood Cancer J. 2022;12(11):161.
- Mato AR, et al. N Engl J Med. 2023;389(1):33-44.
- Montoya S, et al. Cancers (Basel). 2023;15(14):3648.
- Siddiqi T, et al. Lancet. 2023;402(10402):641-654.
- Siddiqi T, et al. Presented at: ASH Annual Meeting; December 10, 2023; San Diego, CA.
- Tambaro FP, et al. J Exp Pharmacol. 2021;13:923-935.
- Calquence tablets [package insert]. AstraZeneca Pharmaceuticals; 2024.
- Calquence capsules [package insert]. AstraZeneca Pharmaceuticals; 2023.
- Brukinsa [package insert]. BeiGene USA; 2024.
- Imbruvica [package insert]. Pharmacyclics; 2024.
- Burger JA, et al. N Engl J Med. 2015;373(25):2425-2437.
- Burger J, et al. Presented at: EHA 2024 Congress; June 2024; Madrid, Spain.
- Moreno C, et al. Lancet Oncol. 2019;20(1):43-56.
- Moreno C, et al. Haematologica. 2022;107(9):2108-2120.
- Shanafelt TD, et al. N Engl J Med. 2019;381(5):432-443.
- Shanafelt TD, et al. Blood. 2022;140(2):112-120.
- Robak T, et al. Curr Med Res Opin. 2024;40(8):1369-1378.
- Ghosh N, et al. Clin Lymphoma Myeloma Leuk. 2024;24(9):e301-e313.
- Woyach JA, et al. N Engl J Med. 2018;379(26):2517-2528.
- Woyach JA, et al. Blood. 2024;143(16):1616-1627.
- Sharman JP, et al. Lancet. 2020;395(10232):1278-1291.
- Sharman JP, et al. Leukemia. 2022;36(4):1171-1175.
- Sharman JP, et al. Presented at: ASH Annual Meeting; December 10, 2023; San Diego, CA.
- Tam CS, et al. Lancet Oncol. 2022;23(8):1031-1043.
- Al-Sawaf O, et al. Blood. Published online July 10, 2024. doi:10.1182/blood.2024024631
- FÜrstenau M, et al. Lancet Oncol. 2024;25(6):744-759.
- Hallek M, et al. Lancet. 2010;376(9747):1164-1174.
- Badoux XC, et al. Curr Hematol Malig Rep. 2011;6(1):36-46.
- Center for Drug Evaluation and Research. Approval package for application number 205552Orig1s01. 2014. Accessed November 8, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2017/205552Orig1S001.pdf
- Byrd JC, et al. N Engl J Med. 2014;371(3):213-223.
- Farooqui MZ, et al. Lancet Oncol. 2015;16(2):169-176.
- Allan JN, et al. Br J Haematol. 2022;196(4):947-953.
- Del Gaizo Moore V, et al. J Clin Invest. 2007;117(1):112-121.
- Rhodes JM, et al. Curr Oncol Rep. 2022;24(12):1705-1713.
- Byrd JC, et al. Blood. 2016;127(1):79-86.
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Key Treatment Principles and Recent Updates In the Management of Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma: Part 1
Key Treatment Principles and Recent Updates In the Management of Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma: Part 1