Less than half of ICUs nationwide follow standardized sedation and analgesia protocols, and less than a third practice daily sedation interruption. Knowing why those care gaps exist—and how to close them with practical strategies—was the focus of an expert panel at the ASHP Midyear 2024 Clinical Meeting & Exhibition, in New Orleans.
The first step is to know where to turn to for tried-and-true practice guidelines, noted Sandra Kane-Gill, PharmD, a professor of pharmacy and therapeutics at the University of Pittsburgh School of Pharmacy. Implementing the evidence-based Pain, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption (PADIS) guidelines for sedation is a good place to start, Dr. Kane-Gill told attendees. Published by the Society of Critical Care Medicine in 2018 (Crit Care Med 2018;46[9]:e825-e873), these guidelines include 37 recommendations across five guideline sections. Several sections are particularly helpful, including “an updated statement around sedation protocols, stating that in critically ill, intubated adults, daily sedation interruption and targeted sedation can achieve and maintain a light level of sedation,” she said. “We want light sedation because evidence shows that it contributes to decreasing mechanical ventilation, decreases ICU length of stay [LOS] and decreases delirium.”
However, as noted, “surveys have shown that only about 44% of ICUs in the U.S. have some type of sedation protocol, and around 30% of the time there was no daily sedation interruption occurring,” Dr. Kane-Gill said (Crit Care Med 2009;37[3]:825-832).
Several systematic institutional barriers explain, in part, why protocol uptake has not been significantly higher, even more than 15 years after that study was conducted, Dr. Kane-Gill noted. “Some surveys say lack of acceptance is one factor; clinicians may feel that the protocol isn’t applicable to all or most of their patients,” she said. “Others [cite] the associated workload, with many pressures on nurses’ time.” It also can be difficult to use some of these tools in sedated, nonverbal and/or non–English-speaking patients, Dr. Kane-Gill added. “And I think everyone can relate to this one: patient assessment fatigue. This is just one more thing we need to ask the patient about. But we need to know how to overcome those barriers, because not doing these assessments is not in the best interest of the patient.”
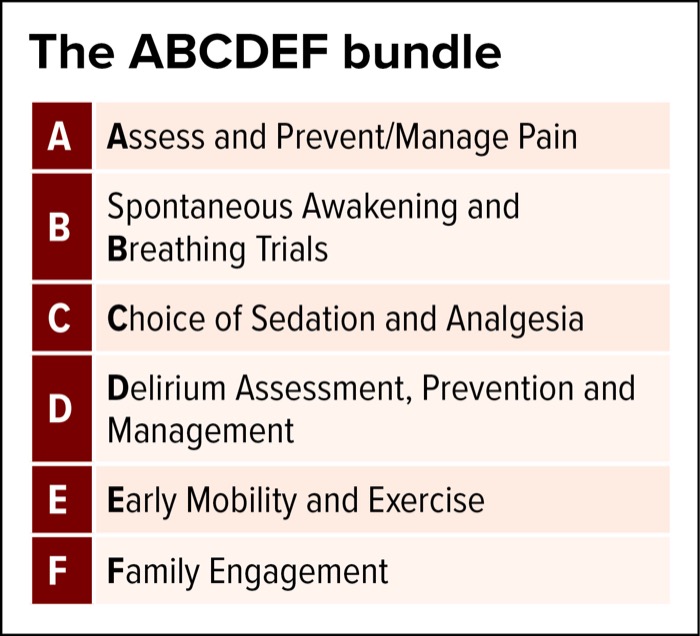
The “ICU Liberation Bundle,” also known as the ABCDEF bundle, has been intentionally developed to ease implementation of the PADIS guidelines (box).
In a study published in Critical Care Explorations in January 2024, the ICU Liberation Bundle reduced ICU LOS by 0.5 days (P=0.02), duration of mechanical ventilation by 0.6 days (P=0.01), and ICU LOS greater than or equal to seven days by 18.1% (P<0.01; 2024;6[1]:e1001).
A 2017 study involving seven California community hospitals found that for every 10% increase in total bundle compliance, patients had a 7% higher odds ratio of hospital survival (OR, 1.07; P<0.001), and for every 10% increase in partial bundle compliance, patients had a 15% higher hospital survival (OR, 1.15; P<0.001; Crit Care Med 2017;45[2]:171-178).
“How can you make implementation of the ICU Liberation Bundle successful?” Dr. Kane-Gill asked. “Start by using an interprofessional team to engage leaders. You want to make this a priority and to have a cultural change within the ICU.” However, she cautioned not to “go in tomorrow and put the whole bundle in place. Work toward small changes, measure them and make sure you are having an effect. You need to be providing multimodal education, with different ways for learners to engage, and also share success stories, which are motivators for any healthcare professional. And of course, you need auditing and feedback. You put the bundle in place, but how often do you actually use it?”
Pharmacists Make a Difference
Pharmacist-led implementation strategies play an important role in improving adoption of the ICU Liberation Bundle and PADIS guidelines, said Mitchell Buckley, PharmD, the director of medical affairs at Innoviva Specialty Therapeutics, who previously served as a clinical pharmacy specialist in the medical ICU at Banner - University Medical Center in Phoenix, for nearly 15 years. “There are many problem-solving opportunities to put pharmacy in the driver’s seat to lead that change.”
In a 2023 review in the Journal of the American College of Clinical Pharmacy, Drs. Buckley, Kane-Gill and colleagues reviewed 12 years of data from randomized controlled trials, quasi-experimental or observational studies comparing pharmacist-led interventions aimed at PADIS management best practices. In four of nine studies, pharmacist-led interventions were associated with a significant reduction in mechanical ventilation duration ranging from 4.0 to 8.5days compared with approximately 5.6 to 14.0days in the control groups.
Three of six studies evaluating LOS showed significant reductions with the intervention group compared with controls. The pharmacist-led intervention group also was associated with significant reductions in opioid analgesia and sedative utilization (J Am Coll Clin Pharm 2023;6[9]:1041-1052).
Some of the strategies used in the review included:
- target pain/sedation goals and agent choice/doses;
- a dexmedetomidine stewardship program;
- overnight tele-ICU PharmD;
- PharmD sedation and delirium assessment;
- education and protocol compliance; and
- clinical decision support trigger alerts.
Dr. Buckley cited an example from the review of an institution that had issues with delirium in their ICU patients. “One of the pharmacy solutions was to develop criteria for a trigger alert that would help them identify at-risk patients, which included advanced age, receiving a benzodiazepine or an antipsychotic,” he said. “The alert would fire to the pharmacist to pay a little more attention to ensure that they were optimizing sedation.”
Another example, he noted, was a dexmedetomidine stewardship program. “They were laser-focused on this over time, with all ICU pharmacists paying closer attention to the appropriate indications for utilization and the dosing strategy,” he said. “Your institution may not have a dexmedetomidine problem, but maybe keep the strategy as an opportunity but change the scenario.” At Banner, “we had a serious problem with dosage creeping up on continuous-infusion fentanyl,” so that became a core focus “on just attacking that one thing before tackling other issues.”
For those still struggling to improve sedation protocol adoption, other resources can help, Dr. Kane-Gill noted, including the APEASE rubric (acceptability, practicability, effectiveness, affordability, side effects and equity), along with pharmacist-led ICU sedation optimization initiatives.
The sources reported no relevant financial disclosures.
This article is from the April 2025 print issue.