Immunoglobulin National Society
Calabasas, California
Advanced Infusion Care
Dallas, Texas
Immunoglobulin National Society
Calabasas, California
Immunoglobulin (Ig) therapy is a highly complex, resource-intensive specialized therapy used to manage a wide range of immune-mediated conditions, including primary and secondary immunodeficiencies, autoimmune diseases, and inflammatory disorders. The increasing complexity of indications and risk factors, individualized dosing strategies, product variability, and infusion-related adverse events demands a high level of clinical expertise for safe and effective treatment.1,2
The success of Ig therapy and optimal patient outcomes are heavily dependent on the competence of the clinical team delivering care. Nurses and pharmacists at the forefront of Ig therapy must possess role-specific knowledge and critical decision-making skills, because their interventions directly influence therapeutic outcomes, patient safety, and continuity of care.2-5
To meet the clinical and operational demands of providing Ig therapy, healthcare organizations must implement structured training pathways, ongoing competency validation, and professional certification.3-6 The Immunoglobulin National Society (IgNS), a professional organization dedicated to advancing the clinical practice of Ig therapy, has addressed this need through the development of evidence-based practice standards and nationally recognized certification programs. These include the Immunoglobulin Certified Nurse (IgCN) and Immunoglobulin Certified Pharmacist (IgCP) credentials, which validate clinical proficiency and provide a framework for ongoing professional development. These programs are developed leveraging robust role delineation studies and nationally recognized psychometric standards, and the examinations deliver a rigorous, legally defensible confirmation of a clinician’s knowledge and readiness to provide Ig therapy.2,7
This article examines the clinical and organizational case for establishing structured and certified teams in Ig therapy with foundational training, ongoing competency assessment, and continuing education to promote clinical precision, patient-centered care, and operational sustainability.
Clinical Rationale for Competency-Based Staffing
Ig therapy involves patient-specific risk assessments, weight- and indication-based dosing, product selection across multiple brands and formulations, and the potential for acute or delayed adverse drug reactions. These complexities demand far more than general infusion competence and require a specialized knowledge base, as well as a coordinated, multidisciplinary approach to providing care.2,3,5
Substandard documentation signals deeper gaps in infusion competency. When clinicians are not adequately trained, they can miss subtle changes in vital signs, bypass validated titration or premedication protocols, and omit critical fields when documenting in the patient record. These omissions may mask emerging risks, delay corrective action, and increase the probability of infusion-related adverse events.2,6
Vague or overlapping role definitions introduce additional vulnerabilities. Without clear accountability, interdisciplinary communication breaks down, treatment initiation stalls, and continuity of care suffers. These breakdowns can compromise patient safety, erode prescriber confidence, and strain relationships with payors.2,6
By contrast, competent staffing directly contributes to efficient clinical decision-making, safer infusions, and higher-quality care. Nurses proficient in Ig therapy are equipped to assess patient risk factors, manage infusion rate changes, deploy appropriate interventions, respond to early signs of clinical intolerance, and provide comprehensive patient counseling. Pharmacists play a particularly important role in verifying diagnosis- and patient-specific dosing regimens, evaluating comorbidities, mitigating adverse interactions, managing lot tracking, and guiding therapeutic interchange when formulary restrictions are applied. Their engagement in patient-specific therapy planning ensures a high degree of safety and regulatory compliance, especially when working in tandem with Ig nurses and prescribers.2,3,6,8
Effective Ig therapy begins with a focused patient assessment. The prescriber, pharmacist, and nurse review the patient’s disease history, comorbidities, and previous exposure to Ig or other biologics to select the appropriate product, dose, and route. The resulting treatment plan includes details on titration, monitoring, patient or caregiver education, and expected outcomes. Ongoing interdisciplinary communication ensures that the plan of care remains current and facilitates coordinated, high-quality care.2,3,6,8
Barriers to Certification
Despite the recognized value of specialty certification in Ig therapy, employers and clinicians alike encounter structural, organizational, and perceptual barriers that limit adoption.9-11
Clinicians often have limited preparation time, uncertainty about testing procedures, and anxiety surrounding high-stakes exams, particularly when formal institutional support is lacking. Early-career staff may not yet meet the 1,500-hour infusion experience requirement, while late-career clinicians may perceive minimal benefit due to impending retirement or discomfort with standardized testing.7,9-13
To mitigate these challenges, some organizations embed certification into clinical ladder programs, pair it with mentorship, and offer protected study time or recognition incentives—approaches that enhance certification uptake, workforce retention, job satisfaction, and role clarity.1,12,14
Consistent with national quality initiatives such as the Centers for Medicare & Medicaid Services Hospital-Acquired Condition Reduction Program, which penalizes hospitals in the worst-performing quartile for preventable harm, certified staff play a pivotal role in reducing adverse events and ensuring compliance with national quality benchmarks.15
Building High-Functioning, Collaborative Teams
The IgNS Standards of Practice emphasize the need for a multidisciplinary care model that integrates all Ig clinicians, including pharmacists, nurses, and prescribers, as a cohesive team to ensure the safe, consistent, and patient-centered delivery of Ig therapy across diverse clinical settings. Each care team member must meet competency standards, and their roles should be clearly defined and regularly re-evaluated to reflect evolving products, indications, practice standards, and regulatory expectations. 2,6
Effective Ig therapy teams arise from deliberate hiring, systematic competency validation, and cross-disciplinary collaboration. Consistent with IgNS standards, organizations should confirm clinician competence at hire and at scheduled intervals, using objective, Ig therapy–specific metrics that go beyond licensure. Routine reassessment is required to align with evolving evidence, standards, and regulatory requirements.2,6
Once competencies are confirmed and clinicians are integrated into the team, a structured onboarding process is prudent, including mentorship, simulation-based training, and supervised practice in administering Ig therapy, in accordance with the IgNS practice standards. These components are particularly important in settings with high patient acuity or frequent transitions of care.2,6,8,12
Ig Certification as a Framework For Clinical Excellence
The dynamic and highly complex nature of Ig therapy, evolving therapeutic indications, and patient safety risks demand specialized expertise and a continuous education process to support safe clinical practice.2,7 IgNS offers the only nationally recognized certification programs for nurses and pharmacists involved in Ig therapy: the IgCN and IgCP credentials that provide an essential framework for demonstrating, validating, documenting, and maintaining clinical competence in this high-risk therapeutic domain.
These credentialing programs are designed to assess the knowledge, clinical reasoning, and technical skills required to safely manage Ig therapy across care settings. Certification is based on formal practice analysis studies and psychometrically validated examination content, ensuring that credential holders meet nationally accepted standards for specialized Ig therapy competence. The examinations evaluate knowledge of Ig products, indications, risk mitigation strategies, patient assessment, dosing principles, adverse reaction management, and interdisciplinary collaboration.
The benefits of certification in Ig therapy extend across multiple levels of the healthcare system, including clinicians, employers, and patients (Table 1). Overall, Ig credentialing supports clinical competence, professional development, and system-wide quality improvement, and drives the development of trust, safety, and excellence in Ig therapy delivery.
| Table 1. Stakeholder-Specific Benefits Of IgCN and IgCP Certifications |
| Clinicians |
|---|
Becoming an IgCN/IgCP is the only method of validation and recognition of the specialized knowledge, skills, and expertise of an Ig nurse or Ig pharmacist. In addition, IgCN/IgCP certification:
|
| Employers |
|
| Patients? |
|
| Based on reference 7. |
Eligibility criteria and examination structure
To qualify for certification, candidates must7:
- hold a current, unrestricted license as a registered nurse or registered pharmacist in the United States or Canada; and
- document a minimum of 1,500 hours of relevant experience in Ig therapy within the previous 2 years. This may include practice in direct patient care, managed care, education, research, or the pharmaceutical/device industry.
The credentialing examination is offered during four 2-week windows per year with in-person or remote-proctored formats. The 2-hour exam consists of 110 multiple-choice questions aligned with core content domains in Ig nursing or pharmacy.
For more information, visit ig-ns.org/certification/ and download the free Candidate Handbook.2
Core competency domains in Ig therapy practice
Certification provides a formal validation of expertise, but maintaining clinical excellence in Ig therapy requires ongoing education and structured training. A comprehensive staff competency program is essential for ensuring that nurses, pharmacists, and other healthcare professionals are equipped to manage the nuanced clinical, operational, and safety considerations involved in Ig therapy delivery.2
According to the IgNS Standards of Practice Edition 3.2, all organizations engaged in Ig therapy must implement standardized training protocols that address both foundational knowledge and evolving clinical practices. Competency must be established at onboarding and routinely reassessed, particularly when changes occur in product availability, indications, or regulatory guidelines. The training should also be role-specific and tailored to each clinician’s scope of practice.2
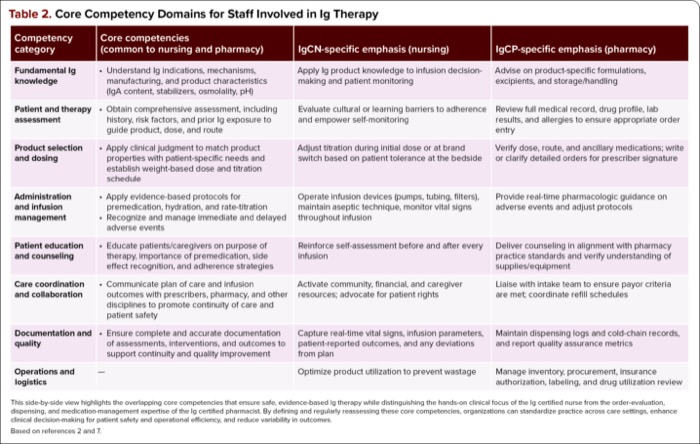
Table 2 summarizes key areas that should be covered in a comprehensive Ig therapy training program, including general core competencies and discipline-specific skills for nurses and pharmacists.2
| Table 2. Core Competency Domains for Staff Involved in Ig Therapy | |||
| Competency category | Core competencies (common to nursing and pharmacy) | IgCN-specific emphasis (nursing) | IgCP-specific emphasis (pharmacy) |
|---|---|---|---|
| Fundamental Ig knowledge |
| Apply Ig product knowledge to infusion decision-making and patient monitoring | Advise on product-specific formulations, excipients, and storage/handling |
| Patient and therapy assessment |
| Evaluate cultural or learning barriers to adherence and empower self-monitoring | Review full medical record, drug profile, lab results, and allergies to ensure appropriate order entry |
| Product selection and dosing |
| Adjust titration during initial dose or at brand switch based on patient tolerance at the bedside | Verify dose, route, and ancillary medications; write or clarify detailed orders for prescriber signature |
| Administration and infusion management |
| Operate infusion devices (pumps, tubing, filters), maintain aseptic technique, monitor vital signs throughout infusion | Provide real-time pharmacologic guidance on adverse events and adjust protocols |
| Patient education and counseling |
| Reinforce self-assessment before and after every infusion | Deliver counseling in alignment with pharmacy practice standards and verify understanding of supplies/equipment |
| Care coordination and collaboration |
| Activate community, financial, and caregiver resources; advocate for patient rights | Liaise with intake team to ensure payor criteria are met; coordinate refill schedules |
| Documentation and quality |
| Capture real-time vital signs, infusion parameters, patient-reported outcomes, and any deviations from plan | Maintain dispensing logs and cold-chain records, and report quality assurance metrics |
| Operations and logistics | — | Optimize product utilization to prevent wastage | Manage inventory, procurement, insurance authorization, labeling, and drug utilization review |
| This side-by-side view highlights the overlapping core competencies that ensure safe, evidence-based Ig therapy while distinguishing the hands-on clinical focus of the Ig certified nurse from the order-evaluation, dispensing, and medication-management expertise of the Ig certified pharmacist. By defining and regularly reassessing these core competencies, organizations can standardize practice across care settings, enhance clinical decision-making for patient safety and operational efficiency, and reduce variability in outcomes. Based on references 2 and 7. | |||
Significance of Certification for Organizations
The IgNS Standards of Practice recommend that certification be embedded within broader clinical governance frameworks, serving as a mechanism to validate clinician expertise and support quality assurance. When combined with structured onboarding, product-specific training, and ongoing competency evaluation, certification promotes professional accountability, clinical confidence, and system-wide excellence.2
Certified clinicians are more likely to follow evidence-based protocols, recognize early signs of infusion-related complications, reduce the risks for clinical sequelae, and coordinate care across disciplines. These capabilities reduce preventable adverse events, inappropriate product use, and therapy discontinuation. In value-based care models, such improvements translate directly into operational efficiency and payor confidence.2,3,6
Organizations that prioritize staff certification often gain tangible benefits, including enhanced workflow consistency, stronger relationships with prescribers and payors, and improved patient outcomes. Certified teams also reduce inefficiencies associated with staff turnover, training delays, and onboarding costs—factors that are particularly critical in home and specialty infusion programs.4,16
Beyond system performance, certification contributes to clinician satisfaction and retention. Professionals who are supported in their development are more likely to remain engaged, serve as mentors, and foster a culture of excellence.1,9,12,17 Ultimately, certification is a strategic investment in the organization’s Ig therapy programs. A competent, certified clinical team and national Ig therapy standards-based policies elevate care quality, enable scalability, and reinforce institutional credibility.4,16
Conclusion
Ig therapy is a high-risk, high-variability intervention that requires consistent clinical oversight, product-specific knowledge, and individualized patient management. Expanding indications and care settings of Ig therapy demand not only individual clinician competence but organizational commitment to multidisciplinary excellence.
As emphasized in the IgNS Standards of Practice Edition 3.2, optimal management of Ig therapy requires a collaborative, well-trained, and certified care team. This includes not only foundational education and competency testing but also structured retraining when changes occur in Ig products, indications, or standards of care. The IgCN and IgCP certifications offer a nationally recognized pathway to validate clinical readiness and reinforce adherence to best practices. When integrated into institutional policy, these credentials contribute to measurable improvements in patient safety, workflow efficiency, and prescriber and payor trust. Overall, strategic investment in competency-based staffing, professional certification, and continuing education is essential for safe, effective, and sustainable Ig therapy programs.
The authors reported no relevant financial disclosures.
References
- Wei H, et al. Int J Nurs Sci. 2023;10(2):189-198.
- Immunoglobulin National Society. Immunoglobulin Therapy Standards of Practice. Edition 3.2. Accessed June 12, 2025. https://ig-ns.org/product/ig-therapy-standards-of-practice-version-3-2/
- Halm MA. Am J Crit Care. 2021;30(2):156-160.
- Board of Pharmacy Specialties. The role of board certification in patient safety. Published March 2025. Accessed June 12, 2025. https://bpsweb.org/2025/03/18/the-role-of-board-certification-in-patient-safety/
- LaNou E, et al. Am J Health Syst Pharm. 2025;82(6):306-316.
- Kennedy-Metz LR, et al. J Thorac Cardiovasc Surg. 2022;163(3):1096-1104.
- Immunoglobulin National Society. IgCN® and IgCP® certification information. Accessed June 12, 2025. https://www.ig-ns.org/certification/
- National Home Infusion Foundation. Home–infusion nurse workforce study. Infusion Journal. 2024;3(2):35–43.
- Dierkes AM. J Nurs Adm. 2021;51(5):249-256.
- Alsayed RA, et al. Front Med (Lausanne). 2025;12:1528856.
- Zdunek K, et al. BMC Med Educ. 2024;24:1169.
- Gill–Bonanca K. Am Nurse J. 2024;19(8).
- Infusion Nurses Society. CRNI® Examination Handbook. 2024. Accessed June 12, 2025. https://www.ins1.org/wp-content/uploads/2024/04/CRNI-Handbook-FINAL-April-2024.pdf
- Leamon MJ, et al. J Nurs Adm. 2023;53(2):104-109.
- Centers for Medicare & Medicaid Services. Hospital-Acquired Condition (HAC) Reduction Program, Fiscal Year 2025 Fact Sheet. 2024. Accessed June 12, 2025. https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/hospital-acquired-condition-reduction-program-hacrp
- American Nurses Association. Reimagining the Economic Value of Nursing: Summary Report. Published January 2025. Accessed June 12, 2025. https://www.nursingworld.org/globalassets/ancc/research/re-imagining-economic-value-of-summary-report-jan-2025.pdf
- NSI Nursing Solutions, Inc. 2025 National Health Care Retention & RN Staffing Report. Published 2025. Accessed June 12, 2025. https://www.nsinursingsolutions.com/documents/library/nsi_national_health_care_retention_report.pdf
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.
Download to read this article in PDF document:![]() Optimizing Ig Therapy Through Certification and Competency: A Multidisciplinary Imperative
Optimizing Ig Therapy Through Certification and Competency: A Multidisciplinary Imperative