Originally published by our sister publication Specialty Pharmacy Continuum
Two new studies presented at the IgNS 2024 National Conference, in Washington, D.C., offer insight into treatment options for immune globulin (IG) therapy.
Protection Against SARS-CoV-2
In the second study (poster 106), researchers determined that plasma-derived IG infusions for treating PI provide some antibody protection against SARS-CoV-2.
“During the pandemic, we got a lot of questions about the antibody levels in our products,” said Kim Clark, PharmD, MBA, IgCP, the medical affairs excellence and publications lead for Kedrion Bipharma. Besides responding to such questions individually, Kedrion decided to publish data about SARS-CoV-2 antibody titer levels in multiple batches of its IG products.
The company used a neutralizing antibody assay, the GenScript cPass Kit, to detect levels of antibodies capable of binding to the SARS-CoV-2 spike protein. They tested two different IG products licensed to treat PI: Gammaplex 5% (50 g/L) and 10% (100 g/L) (immune globulin intravenous [human] liquid, Bio Products Laboratory).
Of 82 batches tested, 21 were filled in 2021, 35 in 2022 and 26 in 2023; 77 of the 82 batches were Gammaplex 10%. It takes six to nine months between the time someone donates plasma and a Gammaplex solution is available, Dr. Clark noted.
This means some of the 2021 batches were derived from plasma donated in 2020, as the COVID-19 pandemic began and before vaccines were widely available. Antibody levels in these early stages likely were derived from someone’s own immune response to the SARS-CoV-2 virus, eventually supplemented by antibody protection from vaccines.
Levels of SARS-CoV-2 antibodies increased significantly from 2021 to 2022 (P<0.01) but not from 2022 to 2023 (P=0.53). The mean potency of antibodies across all batches was 215 IU/mg immunoglobulin G (IgG); median potency was 213 IU/mg IgG. There were detectable SARS-CoV-2 antibodies in every batch.
“It’s very hard to correlate antibody levels to degree of protection, but we can say with confidence that antibodies are there,” said Martyn Paddick, BSc, who conducted the assays and heads Kedrion’s research and development efforts in the United Kingdom.
This antibody strength of the older batches does not surprise Joe DiStefano, RPh, IgCP, CSP, the vice president for clinical programs at Nufactor, an IG and home infusion provider based in Temecula, Calif. “The older batches were closer to the height of the pandemic. When people are being infected or getting vaccinations their COVID antibodies are spiking,” Mr. DiStefano said.
The sources reported no relevant financial disclosures.
Improving Primary Immunodeficiency
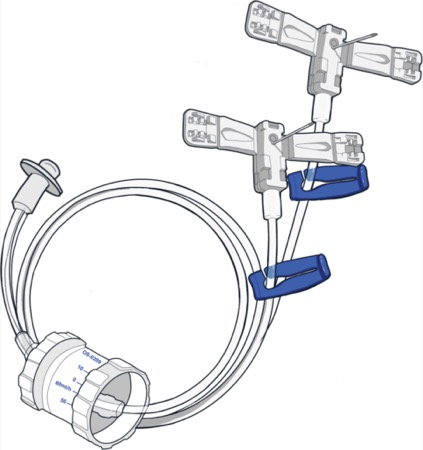
The first study outlined how an FDA-cleared immunoglobulin infusion set from Innovative Health Sciences aims to improve the experience of people with primary immunodeficiency (PI) when self-administering IG therapy.
The OneSett product offers benefits for patients, nurses and pharmacists alike, according to the investigators (poster 107).
Working with other infusion products can be cumbersome, said Melody Bullock, MS, BSN, BS, RN, CRNI, IgCN, a clinical science liaison for Innovative Health Sciences, especially when changing needle size or infusion rates to reduce the risk for site reactions.
“Nurses go back to the pharmacist and say, ‘This isn’t working right. We need different fixed-rate tubing.’ And they were using online calculators to help them decide the right needle sets and right infusion rate to get the infusion in a certain time frame for the patient,” Ms. Bullock said.
Not only is this trial-and-error process tedious and tiresome for patients; it also requires pharmacists to keep more than 150 stock-keeping units (SKUs) on hand to account for every possible tubing and needle combination.
The OneSett product, in contrast, only requires 16 SKUs for possible infusion rates for IG patients. Moreover, it uses a lever instead of a dial, with needles on one end, a flow rate controller in the middle and an extension tubing attached to a syringe. Based on a survey of eight patients, the system is easy to use and supports accurate flow rates, according to the company.
Innovative Health Sciences said it plans to conduct more patient surveys.
Ideally all patients would know the optimal flow ranges for their situation, based on ongoing consultation with a pharmacist and nurse. If so, OneSett could prove helpful.
“This device allows people with CIDP [chronic inflammatory demyelinating polyneuropathy] to adjust their flow rates themselves,” for example, by making the IG flow rate slower to reduce redness or soreness at the injection site, Leslie Myers, PharmD, IgCP, an independent IG therapy expert, told Specialty Pharmacy Continuum. Although OneSett’s ease of use and flexibility are attractive, Dr. Myers noted that some patients will stick with tools they already know.
“You’re not going to be able to convert every patient to this system,” she said, because what they already use works without requiring any new training.
For the pharmacist, this simply means keeping necessary SKUs for other infusion devices, while adding the 16 new SKUs needed for patients who choose to use OneSett. Dr. Myers said she sees this as a minor impact if the benefit is more patient choice.
The sources reported no relevant financial disclosures beyond their stated employment.