Any documentation in the medical record, including all medications, drugs and biologicals, needs to accurately depict patient and treatment complexity. Your goal is to tell the patient’s story accurately and completely and in a codable fashion. Why? Because when the codes are unpacked at the receiving payor, they have to match the severity of the patient’s illness in real life. Is your coding up to this challenge?
Working with your revenue cycle and information technology (IT) team requires timely attention and has a significant impact on revenue and data integrity. Can your team rapidly process a newly approved FDA entity? What are you doing with the plethora of new Healthcare Common Procedure Coding System (HCPCS) codes for 505(b)(2) or Biologics License Application (BLA) Pathways products being released quarterly? Are the folks on your revenue cycle team putting any hard stops to any of these data being transmitted to the payor?
Previously, the Centers for Medicare & Medicaid Services (CMS) would print new information about codes in its MLN Matters publications and include the specifics in tables. However, consistent with CMS’s new approach to reduce “paper work,” only links to the actual data now are being provided. This column is designed to help you make that transition and is based on this MLN Connects newsletter (2024-09-12-MLNC), found at bit.ly/3zKZuRA. I included Tables 1 to 3 in this column; for others, I have provided the actual link.
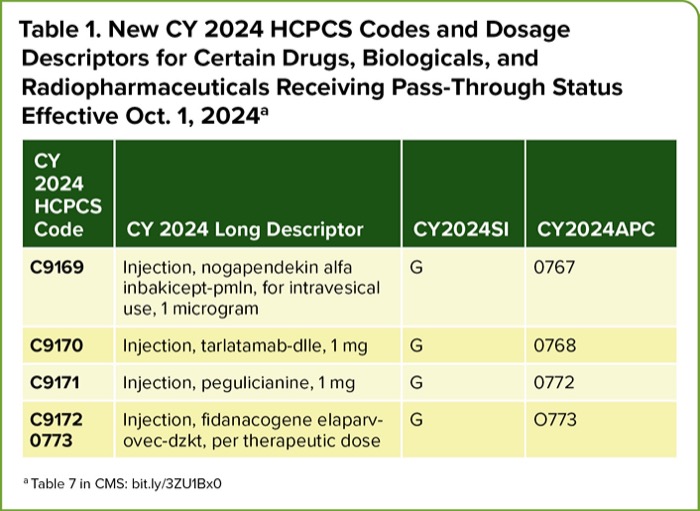
| Table 1. New CY 2024 HCPCS Codes and Dosage Descriptors for Certain Drugs, Biologicals, and Radiopharmaceuticals Receiving Pass-Through Status Effective Oct. 1, 2024a | |||
| CY 2024 HCPCS Code | CY 2024 Long Descriptor | CY2024SI | CY2024APC |
|---|---|---|---|
| C9169 | Injection, nogapendekin alfa inbakicept-pmln, for intravesical use, 1 microgram | G | 0767 |
| C9170 | Injection, tarlatamab-dlle, 1 mg | G | 0768 |
| C9171 | Injection, pegulicianine, 1 mg | G | 0772 |
| C9172 0773 | Injection, fidanacogene elaparvovec-dzkt, per therapeutic dose | G | O773 |
| a Table 7 in CMS: bit.ly/3ZU1Bx0 | |||
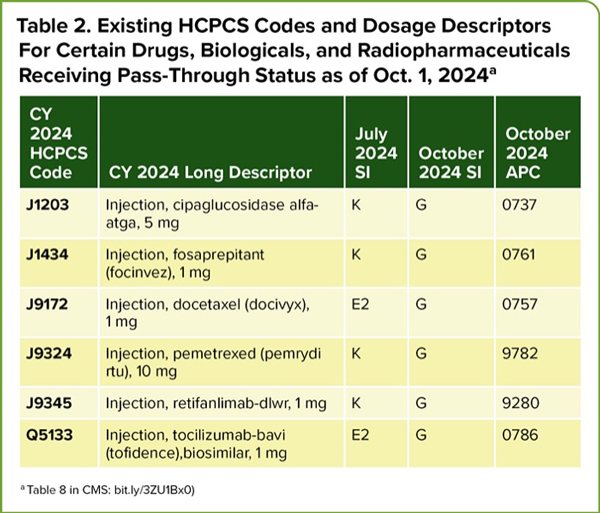
| Table 2. Existing HCPCS Codes and Dosage Descriptors For Certain Drugs, Biologicals, and Radiopharmaceuticals Receiving Pass-Through Status as of Oct. 1, 2024a | ||||
| CY 2024 HCPCS Code | CY 2024 Long Descriptor | July 2024 SI | October 2024 SI | October 2024 APC |
|---|---|---|---|---|
| J1203 | Injection, cipaglucosidase alfa-atga, 5 mg | K | G | 0737 |
| J1434 | Injection, fosaprepitant (focinvez), 1 mg | K | G | 0761 |
| J9172 | Injection, docetaxel (docivyx), 1 mg | E2 | G | 0757 |
| J9324 | Injection, pemetrexed (pemrydi rtu), 10 mg | K | G | 9782 |
| J9345 | Injection, retifanlimab-dlwr, 1 mg | K | G | 9280 |
| Q5133 | Injection, tocilizumab-bavi (tofidence),biosimilar, 1 mg | E2 | G | 0786 |
| a Table 8 in CMS: bit.ly/3ZU1Bx0) | ||||
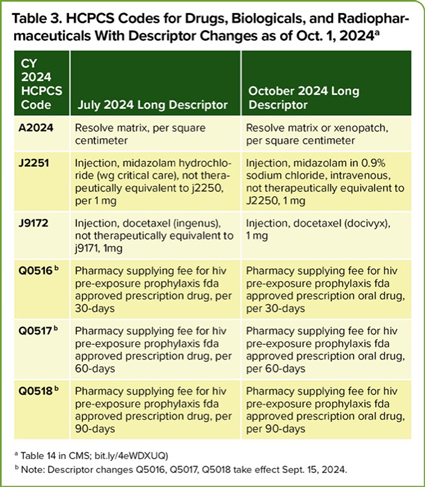
| Table 3. HCPCS Codes for Drugs, Biologicals, and Radiopharmaceuticals With Descriptor Changes as of Oct. 1, 2024a | ||
| CY 2024 HCPCS Code | July 2024 Long Descriptor | October 2024 Long Descriptor |
|---|---|---|
| A2024 | Resolve matrix, per square centimeter | Resolve matrix or xenopatch, per square centimeter |
| J2251 | Injection, midazolam hydrochloride (wg critical care), not therapeutically equivalent to j2250, per 1 mg | Injection, midazolam in 0.9% sodium chloride, intravenous, not therapeutically equivalent to J2250, 1 mg |
| J9172 | Injection, docetaxel (ingenus), not therapeutically equivalent to j9171, 1mg | Injection, docetaxel (docivyx), 1 mg |
| Q0516b | Pharmacy supplying fee for hiv pre-exposure prophylaxis fda approved prescription drug, per 30-days | Pharmacy supplying fee for hiv pre-exposure prophylaxis fda approved prescription oral drug, per 30-days |
| Q0517b | Pharmacy supplying fee for hiv pre-exposure prophylaxis fda approved prescription drug, per 60-days | Pharmacy supplying fee for hiv pre-exposure prophylaxis fda approved prescription oral drug, per 60-days |
| Q0518b | Pharmacy supplying fee for hiv pre-exposure prophylaxis fda approved prescription drug, per 90-days | Pharmacy supplying fee for hiv pre-exposure prophylaxis fda approved prescription oral drug, per 90-days |
a Table 14 in CMS; bit.ly/4eWDXUQ) b Note: Descriptor changes Q5016, Q5017, Q5018 take effect Sept. 15, 2024. | ||
Drugs, Biologicals and Radiopharmaceuticals
Here are links to tables that have important updates for this key class of drugs that your revenue cycle team needs to keep up with.
Existing HCPCS Codes for Certain Drugs, Biologicals, and Radiopharmaceuticals With Pass-Through Status Ending on Sept. 30, 2024
Note that six products move from status indicator (SI) G to SI K. You can access this Table, listed by CMS as Table 9, at bit.ly/3YmQPNx.
Newly Established HCPCS Codes for Drug, Biological, and Radiopharmaceutical as of July 2, 2024
You can access this Table, listed by CMS as Table 10 (1 product), at https://bit.ly/3YmQPNx.
Newly Established HCPCS Codes For Drugs, Biologicals, and Radiopharmaceuticals as of Oct. 1, 2024 Table 11 (34 products)
Note that several of these drugs are “505(b)(2) or Biologics License Application (BLA) Pathways” products. As such, they require delicate coordination between various departments, including the pharmacy acquiring the product, your IT/informaticist and revenue cycle teams, and order entry clinician(s). This coordination is critical because not all products are interchangeable/considered therapeutically equivalent and not all products have the full range of approvals that their branded counterparts sport. Payor requirements/preferences may derail your attempts to reduce drug costs for both your department and the patient. This all has a tremendous impact on order entry. You can access this Table, listed by CMS as Table 11, at bit.ly/3YmQPNx.
HCPCS Codes for Drugs, Biologicals, And Radiopharmaceuticals Deleted as of Sept. 30, 2024
Five products are included in this Table, listed by CMS as Table 12 and accessible at bit.ly/4eWDXUQt.
HCPCS Code for Drug, Biological, And Radiopharmaceutical Changing Payment Status
Two products are included in this Table, listed by CMS as Table 13. It can be accessed at bit.ly/4eWDXUQ.
PrEP for HIV and Related Preventive Services
In a somewhat surprising move, effective Sept. 1, 2024, CMS Medicare moved oral pre-exposure prophylaxis (PrEP) dosage forms out of Part D and into Part B. This means that they will cover oral and injectable forms of PrEP and other related services to prevent HIV without cost sharing (i.e., deductibles or copays) under Medicare Part B. CMS posted a final National Coverage Determination (NCD) for Pre-exposure Prophylaxis (PrEP) Using Antiretroviral Drugs to Prevent Human Immunodeficiency Virus (HIV) Infection (bit.ly/3BRyKiE). Be sure to use these codes for Part B billing.
Health Equity
CMS strives to have its policies support its six Strategic Pillars. These include advancing equity and addressing disparities; expanding access to affordable coverage and care; engaging with partners and communities; driving innovation to provide value-based care; protecting programs for future generations; and fostering excellence in all aspects of CMS operations. Read more details at bit.ly/4eJQBqI.
This article is from the November 2024 print issue.