Originally published by our sister publication Specialty Pharmacy Continuum
The FDA granted a new indication for upadacitinib (Rinvoq, AbbVie) for the treatment of adults with moderate to severe active Crohn’s disease who have not responded adequately to or cannot tolerate one or more tumor necrosis factor blockers.
The indication is supported by data from two induction studies, U-EXCEED and U-EXCEL, as well as the U-ENDURE maintenance study.
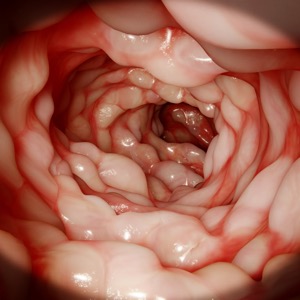
The three phase 3, multicenter, randomized,
