Originally published by our sister publication Specialty Pharmacy Continuum
The FDA granted accelerated approval to mirvetuximab soravtansine-gynx (Elahere, ImmunoGen) to treat adults with folate receptor alpha (FRα)-positive, platinum-resistant epithelial ovarian, fallopian tube or primary peritoneal cancer, who have received one to three prior systemic treatment regimens.
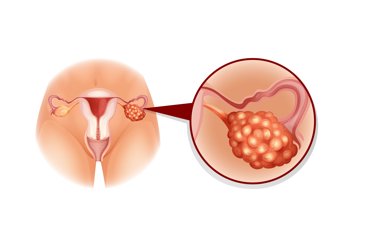
Mirvetuximab soravtansine-gynx is a first-in-class antibody–drug conjugate comprising a FRα-binding
