By PPN News Staff
The FDA extended the time between shots of cabotegravir plus rilpivirine (CAB/RPV; Cabenuva, ViiV Healthcare) to every two months to treat HIV-1 infection in virologically suppressed adults.
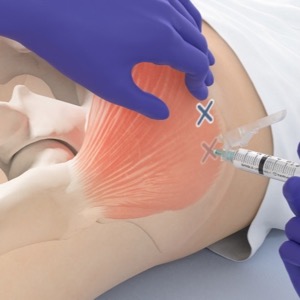
Patients (HIV-1 RNA ≥50 copies/mL) must be on a stable regimen with no history of treatment failure, and without known or suspected resistance to either CAB or RPV.
CAB/RPV, which was approved in January 2021 as a once-monthly treatment for HIV-1 in virologically suppressed adults,
